▎WuXi AppTec Content Team Editor
“On August 9, 2022, Boston-based nonprofit Cure Rare Disease (CRD) announced that the FDA has approved its CRISPR gene-editing therapy called CRD-TMH-001 IND application for the treatment of Duchenne muscular dystrophy (DMD). This therapy is also the first CRISPR gene-editing therapy approved to enter clinical trials for the treatment of DMD.” – Cure Rare Disease press release
Richard Horgan waited three years for this moment.
Richard Hogan, 30, is the founder of Cure Rare Disease. Richard founded the group in 2017 when he was a Harvard Business School student to develop targeted therapies for his younger brother, who has a rare and critical illness.

▲Richard Hogan (picture fromSource: Cure Rare Disease official website)
In 2019, he formed a team of scientists with the University of Massachusetts Medical School, Yale University and other institutions. The FDA-approved therapy for clinical trials is the gene therapy the team tailored for his 27-year-old brother, Terry Horgan. Such therapies are often referred to as N-of-1 therapies because they are developed for only one patient.
This is a special day not only for the Hogan brothers. The therapy also achieved several milestones in the history of medicine: the first personalized CRISPR therapy, the first clinical trial of a gene-editing therapy in muscular dystrophy, a new version of CRISPR (which does not directly change the genetic code itself, but The first clinical test of changing the way cells interpret the genetic code).
A missing piece of DNA, a disease that slowly strips away hope
Terry was born and raised in a small town where his favorite thing about playing Star Wars games as a kid was with his brother. Terry has been interested in technology and computers since he was a child, and is extremely gifted in this area. At the age of 8, he single-handedly assembled a computer. Before he went to college, Terry taught himself a lot of computer-related knowledge through the Internet and video sites. Subsequently, he decided to continue his studies in information science at university. Now, Terry works as an administrative assistant in Cornell’s Information Science Department, as well as an IT staff member for Cure Rare Disease.

▲Terry Hogan (picture fromSource: Cure Rare Disease official website)
However, Terry grew up not only with his passion for computers, but also with a rare disease called Duchenne Muscular Dystrophy.
Duchenne muscular dystrophy is a rare disease caused by mutations (mostly deletions) in the gene encoding the dystrophin protein, which is essential for maintaining muscle cell performance Normal function is critical. Hundreds of mutations are known to cause the disease, and it affects almost exclusively men. In the United States, there are approximately 12,000 to 15,000 cases of Duchenne muscular dystrophy.
Loss of muscle strength and function due to Duchenne muscular dystrophy typically first appears in preschool-age boys and worsens with age. As the disease progresses, severe skeletal and cardiac muscle damage can lead to a complete loss of the ability to walk in early adolescence, accompanied by worsening of cardiac and respiratory symptoms. By late adolescence, these patients may also lose upper-body muscle function and mobility.
Terry’s condition was also caused by the deletion of a DNA segment of his dystrophin gene. This disease, which slowly deprives people of mobility and muscle function, has sapped Terry’s hopes little by little. From a young age, Terry traveled to New York, Philadelphia and Washington, D.C. to seek medical treatment, but doctors were helpless about his illness. In addition, as he gets older, Terry no longer has the opportunity to participate in clinical trials of gene therapy for DMD, because these clinical trials are usually limited to younger boys. For a long time, Terry lost hope for a cure for his illness.
Drugs developed for only one person, decoding the hope of “backup”
However, Terry’s older brother, Richard, persisted. After he founded Cure Rare Disease in 2017 and let his younger brother work in the organization for a period of time, Terry’s heart was planted with the hope of fire again – just a match.
A year later, the story of a little girl named Mila who received her own medicine became the match that ignited the fire in the Hogan brothers.
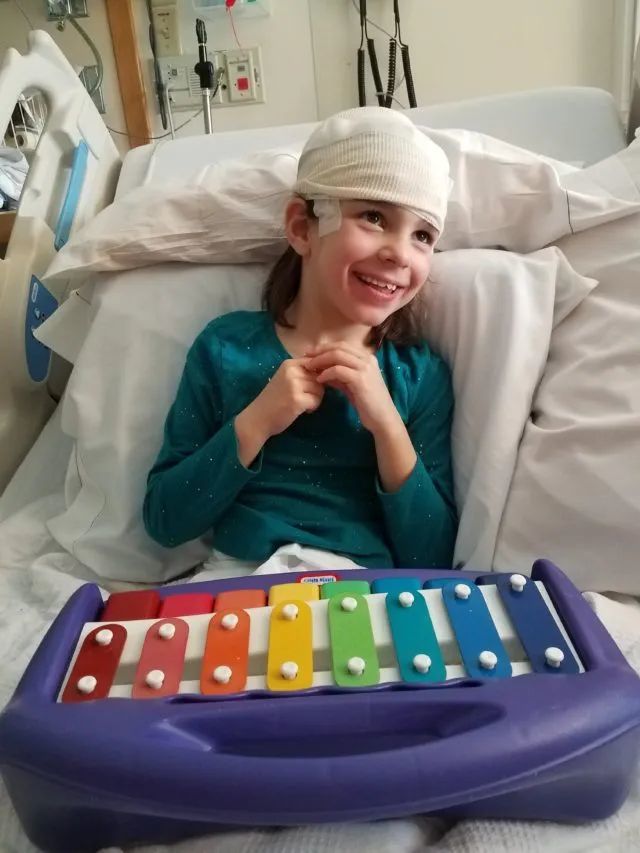
▲Milla (Photo credit: COURTESY JULIA VITARELLO)
Milla also suffers from a rare and deadly genetic disease called Batten disease. Caused by a genetic defect, the disease causes cells to fail to remove the waste they produce, eventually leading to vision loss, ataxia, seizures, developmental decline and even death. After Timothy Yu, a professor of neurology at Boston Children’s Hospital, determined that Mila’s disease was caused by a splicing defect in the CLN7 gene using whole-genome sequencing data, coupled with the mechanism of action of some splicing-regulating antisense oligonucleotide (ASO) drugs at the time Inspired, Professor Timothy Yu pioneered the first drug development program for Mira for only one patient.