Introduction: Existing rabies vaccines target the rabies virus glycoprotein (RABV-G), which is heterogeneous under physiological conditions. Therefore, only a short-term immune response can be produced, which means that the rabies vaccine does not provide life-long protection for the human body. Recently, a research team led by the La Jolla Institute for Immunology in the United States and the Pasteur Institute in France has captured the first high-resolution binding of rabies virus glycoprotein trimers to “pre-fusion” specific neutralizing antibodies. The three-dimensional structure provides a new idea for the design and development of long-acting rabies vaccine.
Untreated rabies infections are nearly 100% fatal, killing 50,000 to 60,000 people each year, many of them children. Human rabies vaccines consist of inactivated virus and only provide complete protection in the short term (6 months to one year of vaccination) and do not elicit lifelong immunity. In humans, vaccine-elicited neutralizing antibody levels typically decline 1 to 5 years after vaccination, so frequent vaccinations are required to maintain neutralizing antibody titers and provide protection against rabies infection. In most low-income countries, where rabies mortality rates are high, it is difficult to afford regular vaccinations and treatment after exposure. Therefore, the development of a long-acting rabies vaccine is critical.
Rabies virus glycoprotein (RABV-G) is the only protein exposed on the surface of the virus and is the target of vaccine-induced neutralizing antibodies. On the viral surface, the structure of RABV-G is heterogeneous, with only a fraction of the identifiable trimers. Structural heterogeneity may affect the production of neutralizing antibodies against the fourth epitope and may result in a shorter time of immune response after vaccination.
In a recent study, a research team led by the La Jolla Institute for Immunology, USA and the Pasteur Institute, France, reported that three proteins that bind to the human antibody RVA122 The structure of aggregated wild-type RABV-G can be used to guide improved vaccine design and identify therapeutic drug targets. The related research results were published in Science Advances under the title of “Structure of the rabies virus glycoprotein trimer bound to a prefusion-specific neutralizing antibody”.

Figure 1 Research results (Source: [1])
Scientists cannot answer precisely why a rabies vaccine does not provide long-term protection, but the heterogeneity of RABV-G structure is a key question. Like a Swiss Army Knife, the RABV-G’s sequence can be unfolded and flipped up when needed. The glycoprotein can move back and forth between prefusion (before fusion with the host cell) and postfusion forms, and it can also fall apart, transforming from a trimeric to a monomeric structure.
This deformation seems to provide an invisibility cloak for the rabies virus, since human antibodies typically only recognize a single site on the protein, and when the protein hides or moves When these protein sites are present, antibodies cannot accurately recognize them and naturally cannot generate an effective immune response.
In this study, researchers combined the glycoprotein RABV-G with the monoclonal antibody RVA122 to identify sites in the viral structure that are vulnerable to antibody attack. RVA122 is the type of antibody required for rabies immunity and can effectively neutralize rabies virus. Binding to RVA122 increases the proportion of RABV-G trimers visible by cryo-electron microscopy by more than 30-fold, enabling high-resolution reconstruction and locking RABV-G in the prefusion conformation, as RVA122 may Neutralizes rabies virus by inhibiting the transition of RABV-G to the postfusion conformation.
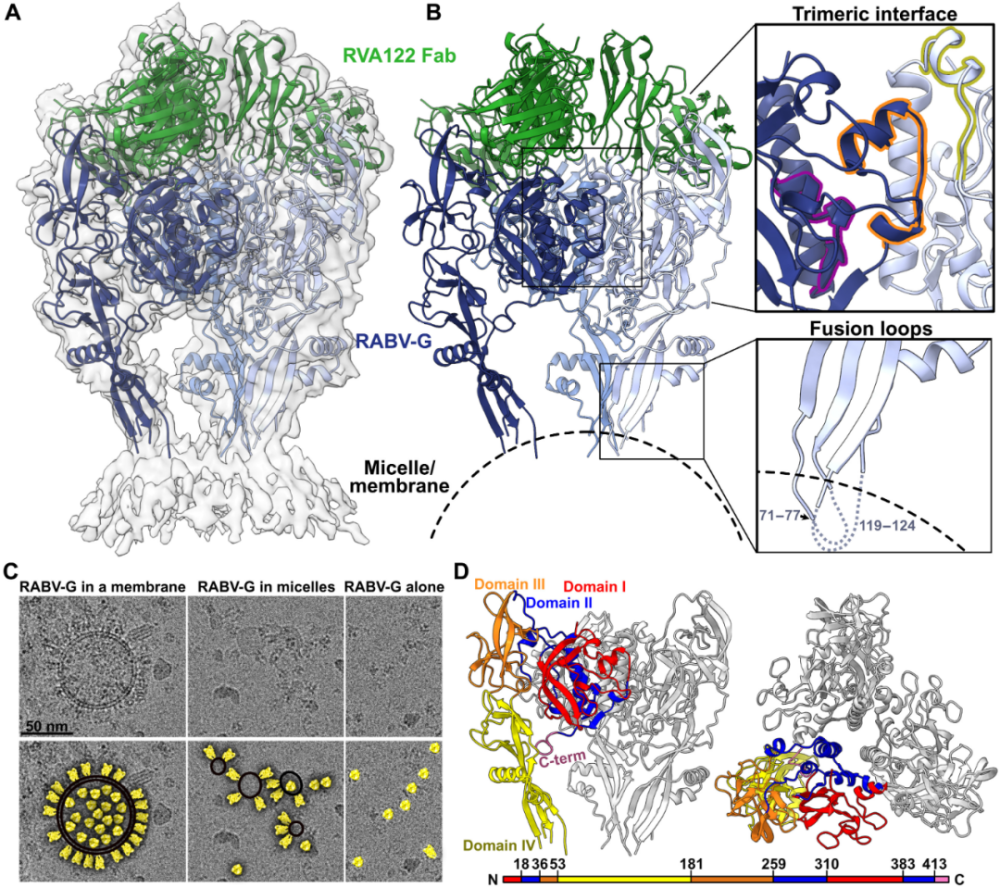

Figure 2 The structure of the prefusion RABV-G trimer bound to neutralizing antibody RVA122 (source: [1])
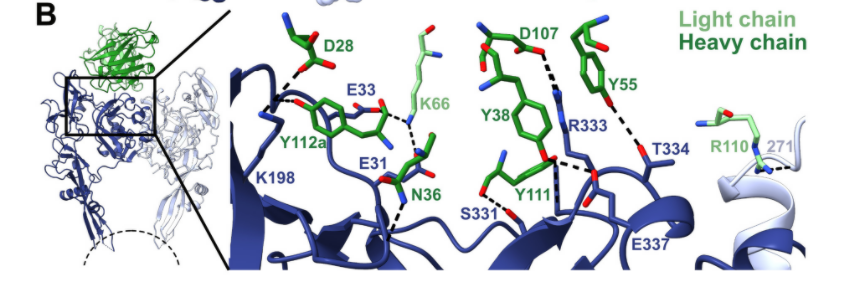
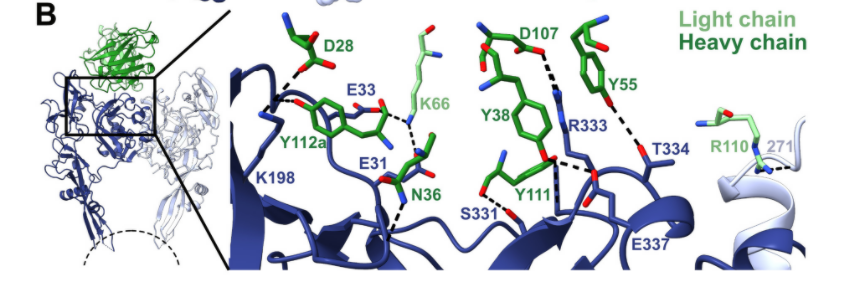
Fig.3 Interaction between RVA122 (green) and RABV-G (dark blue and light blue) (Image source: [1])
Using cryo-electron microscopy equipment, researchers captured high-resolution images of the RABV-G structure, which highlights two key parts of the virus’ structure—the fusion peptide in its native state. form. When the RABV-G trimer is not anchored to the micelle or cell membrane, the fusion peptides are either disordered or interacting. These results suggest that, in addition to driving fusion of the viral and cellular membranes following endosomal acidification, fusion peptides also affect glycoprotein ectodomain binding and trimerization.
To determine whether fusion peptides also affect the conformation and stability of full-length RABV-G, the researchers expressed fusion peptide point mutants as full-length glycoproteins, including the transmembrane domain and cellular Plasmid tails were stained for analysis by flow cytometry and immunofluorescence. All full-length versions of RABV-G mutants were well expressed and readily reached the cell surface, but mutants containing W121A had significantly less full-length protein in the prefusion conformation. This result suggests that for full-length glycoproteins, W121 stabilizes the prefusion conformation by interacting with the membrane, preventing the glycoprotein from transitioning to the alternative conformation. The interaction between W121 and the viral membrane may also stabilize RABV-G on the virion surface.
RABV-G is an important component of vaccines and a target for antibody therapy and potential antiviral drugs, but its structural heterogeneity may lead to suboptimal antibody responses. Trimeric RABV-G before fusion would be an ideal vaccine immunogen, but presentation of this form is challenging due to the instability of the molecule. This study reports the structure of the prefusion trimeric RABV-G, demonstrates the importance of fusion peptides for successful trimerization and conformational stabilization, and visualizes potent and broadly neutralizing antibody epitopes that together guide, Improve rabies vaccine development and post-exposure treatment.
Through this study, scientists have gained a clearer understanding of the structure of the rabies virus and opened the door to the design and development of long-acting rabies vaccines, which may even protect against the entire rabies virus genus. The next step, according to the researchers, is to capture more images of the rabies virus and its relatives, as well as neutralizing antibodies, several of which scientists are working to uncover structures that could shed light on antibody targets shared by rabies viruses.
The study’s lead author said: “Rabies is one of the deadliest viruses we know, and we have lived in its shadow for millennia. However, scientists have The molecular structure on its surface has never been observed. Understanding this structure, like understanding how a virus enters a host cell, is critical for developing more effective vaccines and treatments.”
Writing | Mu Zijiu
Typesetting|Text Competition
References:
[1]Callaway HM, Zyla D, Larrous F, et al. Structure of the rabies virus glycoprotein trimer bound to a prefusion-specific neutralizing antibody. Sci Adv. 2022 Jun 17;8( 24):eabp9151. doi: 10.1126/sciadv.abp9151. Epub 2022 Jun 17. PMID: 35714192; PMCID: PMC9205594.
[2]https:https://medicalxpress.com/news/2022-06-capture-high-resolution-view-rabies-virus.html