This article was written by Karl
Soon, a biological company called Excision BioTherapeutics publicly announced that in its Phase 1/2 clinical trial of EBT-101, the first subject has officially begun to receive Gene therapy for HIV.
The subject was dosed in July 2022, will continue to be monitored for safety, and is expected to be eligible for background antiretroviral therapy (ART) in an assessment of potential cure Analysis of treatment interruption (ATI).

Image source: Screenshot of official news from Excision BioTherapeutics
Shortly after the announcement, the California Institute for Regenerative Medicine (CIRM) announced a $6.85 million grant to support the bio-company to support clinical trials of EBT-101 development.
Associate Professor Isaac Bogoch, a physician-scientist at the University of Toronto, who focuses on AIDS and tropical disease research, gave EBT-101 a high rating of “Brilliant” directly on social networking sites, and said: This technique is accompanied by an easy-to-understand explanation:
“Basically this just snips out the HIV.”
Current HIV treatment: lifelong medication required
HIV has long been a well-known disease in the medical community. The development of antiretroviral therapy (ART), dating back to the 1860s, has saved countless lives afflicted by HIV.
ART makes the diagnosis of AIDS less of a death sentence—some controllers can achieve “long-term remission,” which is life as normal.
HIV borrows the concept of “long-term remission” in leukemia/lymphoma treatment, i.e. functional cure, i.e. not eradicating the virus, but maintaining the reservoir at a lower level, Let the immune system clean up the remaining virus, or strike a balance where the virus doesn’t bounce back.
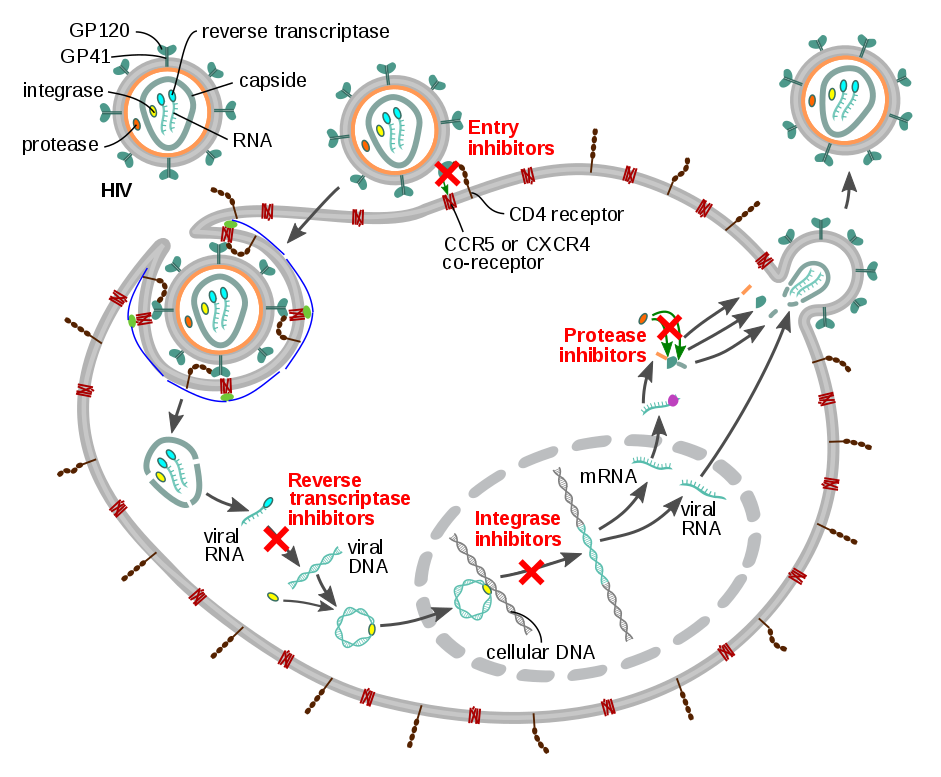
Principle of antiretroviral therapy drugs: inhibition of virus entry, inhibition of protease, inhibition of integrase, inhibition of reverse transcriptase (Source: Wikipedia)
As a result, ART treatment has the problem of “requiring lifelong medication”, even increasing the risk of cumulative comorbidities, and the continued lack of uniform drug access worldwide.
Relevant experts have emphasized the necessity of “HIV cure”, so HIV virus reservoir has become a hot topic of HIV cure.
The HIV reservoir, which is the cell type or anatomical site to which the replicable form of the virus is associated with a more stable dynamic than the main reservoir of actively replicating virus The academic characteristics persist. [1]
A more popular explanation is that the HIV virus synthesizes DNA by reverse transcription, and the DNA is integrated into resting and activated lymphocytes and macrophages. Under the influence of the immune response and antiretroviral therapy, the viral DNA in the cell can stably persist in a dormant-like state. Tissues and cells with similar functions are known as HIV reservoirs.

A highly dispersed reservoir of HIV in the human body
Image source: Reference 2
It is also because of the presence of the viral reservoir that ART does not permanently eliminate the virus and cure the disease caused by the viral infection, and HIV-infected individuals may experience several weeks of ART discontinuation Viral load rebounded.
If the problem with the virus repository can be solved, it might be possible to break through this hurdle.
EBT-101, one cure if successful?
The news of “AIDS cure” is not unfamiliar to everyone.
From first “Berlin patient” to secondA “London patient”, two patients with both AIDS and hematological tumors, after receiving hematopoietic stem cell transplantation from CCR5-delta32 donors, not only was the tumor cured, but HIV was no longer found in the blood.
According to academia, CCR5 is the main receptor for HIV invading T cells, and people with the CCR-delta32 mutation are immune to CCR5-tropic HIV. [3]
However, the high cost, high mortality rate, and strict indications of hematopoietic stem cell transplantation have prevented this method from becoming a mainstream AIDS treatment technology.
And EBT-101 seems to represent a more general possibility – as Dr. Isaac Bogoch says, EBT-101 could snip out the HIV virus Proviruses on repositories.
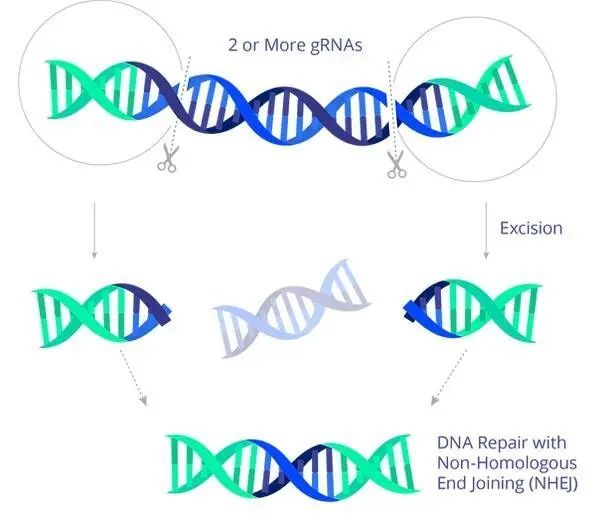
EBT-101 double gRNA excises most of the viral DNA, thereby eliminating viral escape and reproduction.
Image source: Excision BioTherapeutics official website
This magic scissors is the CRISPR-Cas 9 gene editing technology, and the package that delivers the scissors is adeno-associated virus (AAV).
Long-term non-human primate safety data and efficacy data in humanized mice suggest that HIV cure may be possible with this treatment technology. [4]
If you compare MOBA games, “long-term control” is manual reinforcement, “stem cell transplantation” is a plug-in with automatic reinforcement, and “gene editing” is a direct push tower.
In 2019, the discoverers of CCR5, the research group of Professor Deng Hongkui of Peking University, the research group of Chen Hu of the Fifth Medical Center of the PLA General Hospital, and the research group of Wu Hao of Beijing You’an Hospital affiliated to Capital Medical University Collaborated and published a study in ΝΕJM entitled CRISPR-Edited Stem Cells in a Patient with HIV and Acute Lymphocytic Leukemia[5], which reported the use of CRISPR technology to edit CCR5 in hematopoietic stem cells for transplantation to circumvent ethical and safety issues , and pointed out that this method is expected to mass-produce “Berlin patients” and “London patients” to help more AIDS patients.
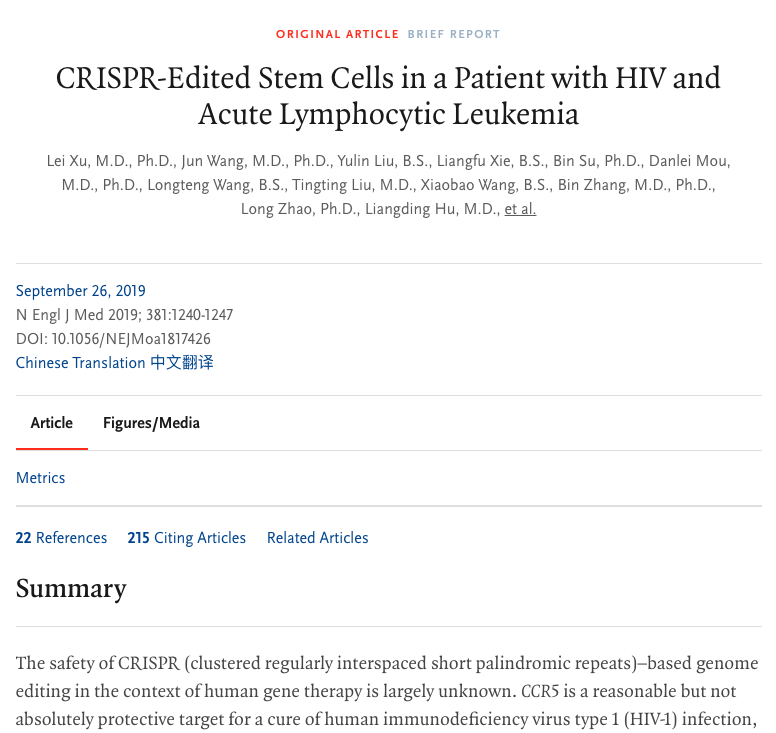
Image source: Reference 5
Although the editing efficiency in the study is not yet high, the safety and potential of this technology have been demonstrated in research.
The proven “Magic Scissors” still to be explored
CIRM President and CEO Maria T. Millan, MD, said, “To date, only a small number of people have been cured of HIV/AIDS, so the proposal to use gene editing to eliminate the virus could be transformative. “
“In California alone, there are nearly 140,000 people living with HIV. HIV infection continues to disproportionately affect marginalized populations, many of whom lack access to drugs to control the virus. Functional healing will have a huge impact on these communities and others around the world.”
With such an optimistic attitude, “Magic Scissors” is still full of challenges while embarking on a new path.
For example, a major problem with implementing CRISPR/Cas9 for gene therapy is the relatively high frequency of off-target effects (OTEs). Cas9’s guide RNA (gRNA) recognizes a 20-bp target DNA sequence, which it binds to and cleaves to “edit” the DNA sequence. However, target sequence binding can tolerate mismatches of up to a few base pairs, which means that there are often thousands of possible binding sites, which can be problematic. [6]
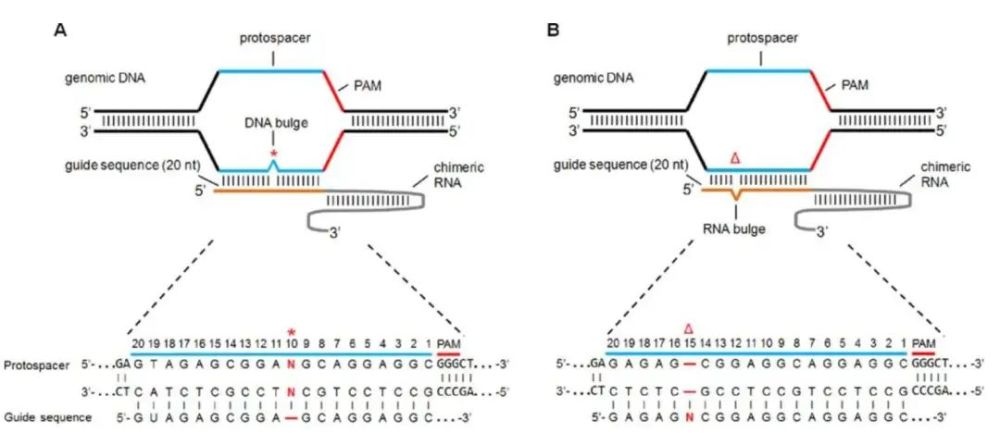
A raised DNA or RNA sequence indicates a mismatch between the leader sequence and the actual cleavage sequence
Image source: Wikipedia
In addition, there are issues of DNA damage toxicity and immunotoxicity. [7]
The good news is that in June 2021, the New England Journal published the results of the first in vivo clinical trial of a CRISPR-Cas 9 gene-editing therapy—a technique in six transthyretin amyloid It is safe and effective in patients with degeneration. [8] also shows that the risk of this technology can be controlled by scientists at a relatively low level.
WhileIf CRISPR-Cas9 is compared to a high point, in fact, the treatment direction of many diseases can have more divergent directions. For example, in the hematology department, which is always fighting for the trend of treatment methods, the same technology is also expected to be applied to the treatment of sickle cell anemia. [9]
Excision BioTherapeutics’ official website also shows that it will try to explore similar treatments in hepatitis B virus and HSV.

Image source: Excision BioTherapeutics official website
From the perspective of a longer period of time, the research fever of gene editing technology is a good thing, and how to explore compliant new treatments based on existing research is a question for contemporary clinicians things to keep going.
Acknowledgments: This article has been professionally reviewed by Li Tongzeng, chief physician of the Department of Infectious Diseases, Beijing You’an Hospital Affiliated to Capital Medical University
Planner: carollero | Producer: gyouza
The source of the title map: Figure Worm Creative
References:
[1]Blankson JN, Persaud D, Siliciano RF. The challenge of viral reservoirs in HIV-1 infection. Annu Rev Med. 2002;53:557-93. doi: 10.1146/annurev .med.53.082901.104024. PMID: 11818490.
[2]Avettand-Fènoël V, Hocqueloux L, Ghosn J, Cheret A, Frange P, Melard A, Viard JP, Rouzioux C. Total HIV-1 DNA, a Marker of Viral Reservoir Dynamics with Clinical Implications. Clin Microbiol Rev. 2016 Oct;29(4):859-80. doi: 10.1128/CMR.00015-16. PMID: 27559075; PMCID: PMC5010749.
[3]Mancuso P, Chen C, Kaminski R, Gordon J, Liao S, Robinson JA, Smith MD, Liu H, Sariyer IK, Sariyer R, Peterson TA, Donadoni M, Williams JB, Siddiqui S, Bunnell BA, Ling B, MacLean AG, Burdo TH, Khalili K. CRISPR based editing of SIV proviral DNA in ART treated non-human primates. Nat Commun. 2020 Nov 27;11(1):6065. doi : 10.1038/s41467-020-19821-7. PMID: 33247091; PMCID: PMC7695718.
[4]https:https://mp.weixin.qq.com/s/xQPXpid_M4HwHvpEKsmtJA
[5]Xu, L., et al., CRISPR-Edited Stem Cells in a Patient with HIV and Acute Lymphocytic Leukemia. New England Journal of Medicine, 2019. 381(13): p. 1240-1247.
[6]Hsu PD, Lander ES, Zhang F. Development and applications of CRISPR-Cas9 for genome engineering. Cell. 2014 Jun 5;157(6):1262-1278. doi: 10.1016/j.cell.2014.05.010. PMID: 24906146; PMCID: PMC4343198.
[7]Uddin F, Rudin CM, Sen T. CRISPR Gene Therapy: Applications, Limitations, and Implications for the Future. Front Oncol. 2020 Aug 7;10:1387. doi: 10.3389 /fonc.2020.01387. PMID: 32850447; PMCID: PMC7427626.
[8]Gillmore JD, Gane E, Taubel J, Kao J, Fontana M, Maitland ML, Seitzer J, O’Connell D, Walsh KR, Wood K, Phillips J, Xu Y , Amaral A, Boyd AP, Cehelsky JE, McKee MD, Schiermeier A, Harari O, Murphy A, Kyratsous CA, Zambrowicz B, Soltys R, Gutstein DE, Leonard J, Sepp-Lorenzino L, Lebwohl D. CRISPR-Cas9 In Vivo Gene Editing for Transthyretin Amyloidosis. N Engl J Med. 2021 Aug 5;385(6):493-502. doi: 10.1056/NEJMoa2107454. Epub 2021 Jun 26. PMID: 34215024.
[9]Ramadier S, Chalumeau A, Felix T, Othman N, Aknoun S, Casini A, Maule G, Masson C, De Cian A, Frati G, Brusson M, Concordet JP, Cavazzana M, Cereseto A, El Nemer W, Amendola M, Wattellier B, Meneghini V, Miccio A. Combination of lentiviral and genome editing technologies for the treatment of sickle cell disease. Mol Ther. 2022 Jan 5;30(1):145 -163. doi: 10.1016/j.ymthe.2021.08.019. Epub 2021 Aug 19. PMID: 34418541; PMCID: PMC8753569.
Lilac Garden is a professional platform for medical practitioners, with the mission of “helping Chinese doctors”. Download the “Lilac Garden App” to discuss cases with peers, study open classes online, use clinical decision-making tools such as medication assistants, and find reliable medical positions in Lilac Talents.