▎WuXi AppTec Content Team Editor
Today,US FDA Announces Approval of Relyvrio (Sodium Benbutate and Taurine Diol Oral Fixed-dose Formulation) Marketed for the treatment of amyotrophic lateral sclerosis (ALS). The approval process for this therapy can be described as twists and turns. In March of this year, the FDA advisory committee voted 6-4, saying that clinical data did not yet support the effectiveness of the therapy. Afterwards, Amylyx submitted further analysis of the clinical trial data and supporting data from other clinical trials. AMX0035 was also first approved by Canadian regulators for conditional marketing.
this September
Second FDA Advisory Committee
On, committee members voted 7-2 in favor of FDA approval of the therapy. Today this therapy finally successfully crossed the finish line!
Amyotrophic lateral sclerosis (ALS) is a progressive neurodegenerative disease that affects nerve cells in the brain and spinal cord, also known as “ALS” “. The motor neurons in the brain and spine of people with the disease continue to die, resulting in muscle weakness and paralysis, ranging from the inability to walk to the inability to speak, swallow, and breathe. People with ALS live an average of only four years after diagnosis. The exact pathogenesis of the disease is currently unknown.
Relyvrio is a combination of two drugs, sodium phenylbutrate and taurursodiol. They can improve the health of mitochondria and endoplasmic reticulum within cells, thereby delaying the death of nerve cells. Preclinical trials have shown that the synergistic effect of the two drugs in combination can reduce nerve cell death due to oxidative stress by 90%. The FDA has granted Relyvrio orphan drug designation and priority review designation.
This approval is based on positive data from a Phase 2 clinical trial that enrolled 137 patients with ALS. The trial met its primary efficacy endpoint of significantly slower decline in motor function in ALS patients treated with Relyvrio at the end of the 6-month randomization period, as measured by the revised ALS Functional Rating Scale.
In addition, follow-up of all randomized subjects for up to 3 years showed that patients who received Relyvrio at the start of the trial were significantly more likely to be in open-label treatment than those who initially received placebo. The risk of death was reduced by 44% (HR 0.56; 95% CI, 0.34-0.92) compared with patients who switched to Relyvrio at the label stage. And the median survival in the group who always received Relyvrio was 6.5 months longer than in the group of patients who initially received placebo (25.0 months vs. 18.5 months).
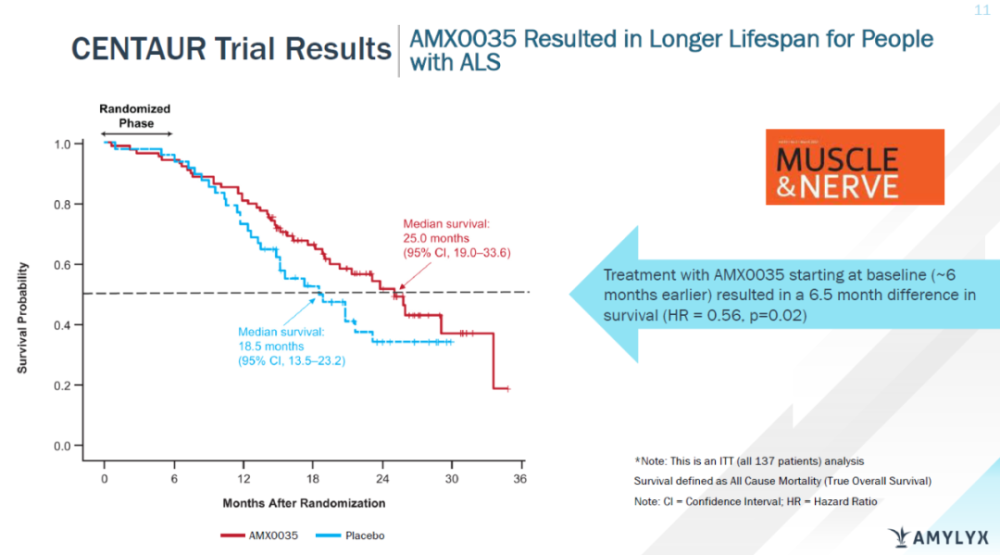
▲Relyvrio prolongs the survival of ALS patients (Image source: Amylyx official website)
Safety, overall, reported adverse events and discontinuation rates were similar between the Relyvrio and placebo groups at 24 weeks, but gastrointestinal events occurred in the Relyvrio group higher rate.
“Amylyx’s goal is to give every patient eligible for Relyvrio the fastest and most efficient way to use their medication, knowing that ALS patients and their families don’t have time for Wait,” said Amylyx co-CEOs Joshua Cohen and Justin Klee, “our team is ready to help ALS patients gain access to this important new treatment option.”
ALS field has received more attention and R&D investment in recent years. According to the statistics of WuXi AppTec’s internal database, as of the end of May this year, nearly 100 ALS candidate drugs have entered the clinical development stage. We look forward to the smooth development of more innovative therapies and their arrival to patients as soon as possible.