On August 9, the FDA released a message on its official website: A sample of the hypoglycemic drug sitagliptin was detected to contain a nitrosamine impurity: Nitroso-STG-19 (NTTP).
Sitagliptin is Merck’s drug for the treatment of type 2 diabetes. Based on sitagliptin, Merck has also developed a combination product of sitagliptin and metformin hydrochloride. These two drugs have brought Merck over $16 billion in revenue over three years.
Nitrosamines are a class of chemical carcinogens. In 2013, Huang Yang, a graduate student of Shanghai Fudan University School of Medicine, was poisoned to death by his roommate. The poison was nitrosamines. According to the prosecution’s allegations at the time, the criminals used at least 30ml of dimethylnitrosamine for poisoning, which was more than 10 times the lethal dose.
Of all the animal experiments with nitrosamines, none of the animals tolerated nitrosamines and did not develop cancer. Therefore, the food and drug industry has always had a zero-tolerance attitude towards nitrosamines.
In 2018, due to the detection of N-dimethylnitrosamine (NDMA) impurities in the valsartan API, the domestic API giant Huahai Pharmaceutical was banned from exporting by the FDA. Exports from China, Japan and other places were also restricted, causing Huahai’s profit to drop to the bottom. On August 11, Huahai Pharmaceutical told the Health and Knowledge Bureau: The lawsuit has not been completed so far.
But this time, the FDA has taken a much more moderate attitude towards the detection of nitrosamines in Merck’s hypoglycemic drugs. The solution given is: Sitagliptin also You can continue to use it, but pay attention to the dosage.
This kind of treatment, the double standard is obvious.
Merck Drugs Contain Nitrosamines: Pay Attention to Dosage
FDA disclosed the problematic sitagliptin, mainly referring to Merck’s patented drug “Jienovi”, but also to its same series of products. will have an impact.
“Genovi” is the world’s first approved oral DPP-4 inhibitor, approved by the FDA in 2006 for the treatment of type 2 diabetes. The response of the drug was good, with sales reaching $668 million in 2007, and its revenue easily surpassed the $1 billion mark within three years of its launch.
Based on the market performance of “Jenovi”, in 2007, Merck developed the sitagliptin metformin “Jenoda”, and subsequently launched a sustained-release dosage form. China’s State Food and Drug Administration approved the listing of “Jenovi” in China in March 2010.
Globally, “Jenovo” is the main source of revenue for this series of sitagliptin drugs. According to Merck’s 2021 annual report, the global sales of “Jenovi” in 2019, 2020 and 2021 were US$3.482 billion, US$3.306 billion and US$3.324 billion respectively. In the three years, the combined revenue of “Jenovi” and “Jenoda” reached 16.088 billion US dollars.
The FDA is also a rat against the topic that sitagliptin contains carcinogens. The drug is in great demand in the U.S. market. FDA expresses concern that the sudden shortage of the drug will affect the lives of patients. It can only recommend that doctors continue to use related drugs in clinical practice, but the dosage needs to be controlled.
In fact, sales of the sitagliptin line of drugs are already declining. In the first half of this year, Merck’s “Jenovia” sales reached US$1.535 billion, a year-on-year decrease of 2%; while “Jenoda” sales also fell by 4%. This is because sitagliptin is facing a patent cliff.
Merck & Co. disclosed in its 2022 interim report that the market exclusivity of the two sitagliptin drugs in the United States will end in January 2023. Merck is actively promoting the entry into force of the “salt/polymorphic patent” for related products, and if it goes well, the patent can be extended to 2027.
On the Chinese side, the compound patent for this drug will expire in July 2022, and all patents will expire in 2024. According to Pharma ONE database data, the sales of sitagliptin preparations in the Chinese market will reach 3.4 billion yuan in 2021.
At present, domestic companies such as Kelun Pharmaceutical, Shijiazhuang Pharmaceutical, Chia Tai Tianqing, Ganli Pharmaceutical and other companies have been approved for sitagliptin phosphate generic drugs. One of the companies told Health Insights that due to patents, the product cannot be sold now. It will also be available in 2024 at the earliest.
Huahai Valsartan Incident: Suppressed by FDA
Merck’s drug was found to have nitrosamine impurities, and it was not immediately removed from the shelves, which is much better than China’s Huahai Pharmaceutical.
In June 2018, Prinston Pharmaceuticals, a US manufacturer of valsartan products, pointed out to the FDA: The chemical nitrodimethylamine (NDMA).
In July 2018, Huahai Pharmaceutical issued an announcement acknowledging the existence of NDMA in the valsartan API, with “very little content”. At the same time, Huahai clearly stated that the relevant APIs “are produced in strict accordance with the GMP standards and registration regulations of the countries where they are supplied. Meets ICH standards.”
Containing nitrosamines in pharmaceuticals was still an emergency at that time, and the registration regulations of various countries did not have a clear standard definition for this impurity caused by the production process.
Huahai Pharmaceutical was a globalThe main supplier of valsartan, after the nitrosamine incident, the United States, the European Union, Japan and other regions have launched strict inspections of valsartan APIs, and Huahai Pharmaceutical has launched a series of related products. Potty was recalled.
FDA has hit hardest. In September 2018, the FDA announced that it was banning the export of all APIs and prepared products made from APIs produced by Huahai Pharmaceutical’s southern Sichuan production base to the United States. Subsequently, the official announcement of the Italian and European Medicines Agency stated that Huahai Pharmaceutical’s valsartan raw materials and intermediates were prohibited from being supplied to the European market.
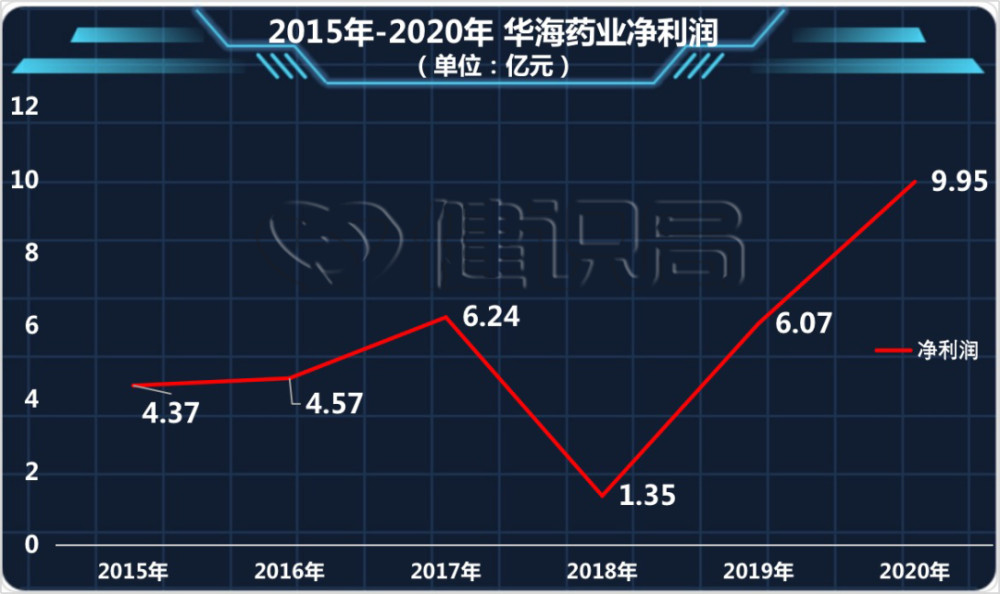
Meanwhile, U.S. consumers began to sue Huahai in court. For a time, Huahai Pharmaceutical became the target of public criticism, and the company’s performance was also seriously affected: since 2015, Huahai Pharmaceutical’s net profit has not been less than 400 million yuan, and in 2017 it has reached 624 million yuan; but In 2018, the net profit plummeted to 135 million yuan.
The State Food and Drug Administration also responded quickly and issued an announcement requiring 5 pharmaceutical companies including Chongqing Kangker Pharmaceutical Co., Ltd. to stop using Huahai Pharmaceutical’s valsartan API, and to comply with regulations Recall related drugs.
In May 2020, the State Food and Drug Administration issued a notice on “Technology for Research on Nitrosamine Impurities in Chemical Drugs until Principles (Trial Implementation)” to further control the safety and quality of drugs,
strong>The direct cause is the Huahai Incident.
In December 2019, Huahai Pharmaceutical’s exports to the EU were resumed; in November 2021, Huahai Pharmaceutical announced that it had received FDA approval for Huahai Pharmaceutical’s raw material drug production base in southern Sichuan. Import ban lifting letter.
The lifting of the injunction does not mean the end of the Valsartan incident. Huahai Pharmaceutical’s many lawsuits in the United States are still ongoing. The 2021 annual report Zhonghuahai Pharmaceutical stated: Due to the involvement of international arbitration, lawyers believe that the procedural processing of this case may take two to three years.
Manufacturers should indeed pay for their faults. However, the difference in the FDA’s attitude towards the raw material manufacturers of sitagliptin and valsartan is too obvious.

Written|Niacinamide
Editing|Jiang Yun Jia Ting
Operations | Twenty-Three
Illustration | Visual China
#FDA##Huahai Pharmaceutical#