Compared with China’s good attitude of “negotiating if you can’t negotiate”, the US medical insurance attitude is tougher, and those who refuse to negotiate may face a sales tax of up to 95%.
The wind of the negotiation of medical insurance collection is blowing to the east coast of the Pacific Ocean.
“Today, the American people won. Some special interest groups lost,” President Biden said of the passage of the Inflation Reduction Act (IRA). JAMA even published an “opinion” pointing out that in the next 10 years, the US version of “medical insurance negotiations” may save the federal government $288 billion.
The implementation goals of the IRA Act can be mainly summarized as “one liter, one drop and one increase”. “One drop” means that the federal government negotiates with pharmaceutical companies to reduce the price of prescription drugs under “Medicare” and reduce medical costs. This is similar to the centralized procurement of medicines in my country.
Some people are happy and others are sad. The Pharmaceutical Research and Manufacturers Association (PhRMA), which represents the interests of pharmaceutical companies, has spent $150 million on public relations and boycotting negotiations.
How much money can people save by seeing a doctor after the medical insurance negotiation?
Comprehensive media coverage, the IRA is a landmark. Because it is the establishment of “Federal Health Insurance”, the United States has passed legislation for the first time to authorize the federal government to intervene in drug prices. There are three core contents:
1. Limit drug price increases: Starting in 2023, the annual increase in drug prices must not exceed inflation. Otherwise, the relevant pharmaceutical companies need to pay the difference to the trust fund under the “Federal Health Insurance”.
2. Set a “cap” on out-of-pocket expenses.
3. Prescription drug price reductions: Authorize the federal government to negotiate prices for the most Medicare-funded drugs in Medicare categories B and D.
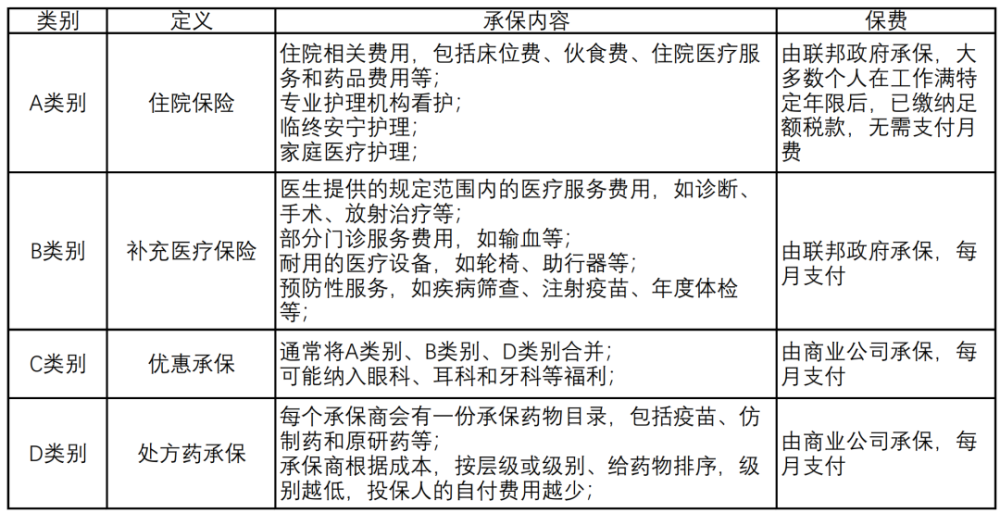
Image description: Medicare has 4 categories A-D. /homemade
According to the White House “Digital News,” the implementation of an IRA could bring real money to the public. Include:
5 million-7 million Medicare policyholders may enjoy cheaper prescription drugs;
The 50 million people who are insured in Category D will have peace of mind “because their out-of-pocket portion of their drug bills cannot exceed $2,000 per year”;
13 million policyholders can save $800 in annual health insurance premiums;
3.3 million policyholders have monthly insulin out-of-pocket costs of no more than $35;
Limit premium increases for D class policyholders in 2024-2030;
Promote more low-income earners with subsidies.
U.S. health care is tough
Refuse to negotiate or face 95% sales tax
Among a series of laws and regulations, drug price negotiation is the foundation and core of “one drop”, and it has received the highest attention. According to the content of the IRA, the US version of “Medical Insurance Negotiation” basically clarifies the “hard indicators” such as the timetable, negotiation objects, negotiation strength, and punishment measures.
From the perspective of time, it will be gradually advanced in 4 stages.
The first negotiating list will be announced on September 1, 2024, effective from 2026, and a total of 10 drugs will be included. The second and third batches of drugs that will take effect in 2027 and 2028 will increase to 15. 20 species by 2029. This means that by 2029, there will be 60 drugs available in the U.S. at negotiated prices.
The first two batches of negotiations are mainly selected from the D category. After the third batch, the inclusion of class B drugs began.
According to the JAMA article, it will be up to the federal government to decide which drugs will be negotiated and haggled. Selection criteria include:
In categories B and D, the top 50 drugs in total expenditure. The JAMA chart shows that in these two categories, the phenomenon that a few drugs occupy most of the medical insurance funds is very prominent;
Small molecule drugs that have been on the market for more than 9 years;
Biologics that have been on the market for more than 13 years;
Original medicine and no generics or biosimilars.
The “markdown criteria” have been clarified:
Listed for 9-12 years, the negotiated price ceiling is 75% of the average price;
Listed for 12-16 years, capped at 65% of the average price;
For more than 16 years, 40%.
Different from my country’s centralized procurement, which “will go away if we can’t reach an agreement”, the IRA has “punishment measures” for companies that refuse to negotiate: high taxes are levied. In the first year, it is levied at 65% of the sales of the previous year, and thereafter it increases to 95% year by year.
If it is negotiated in the early stage but not implemented in the later stage, the company will sufferpenalty. The amount of the fine is calculated based on the difference of 10 times the selling price and the negotiated price.
Pharma companies can also opt out of Medicare to avoid negotiations. Due to the substantial share of prescription drugs in “Medicare”, pharmaceutical companies may not be willing to give up this track.
In 2017, commercial health insurance was the largest payer of prescription drugs at 42%. “Medicare” is the second largest payer, accounting for 30%.
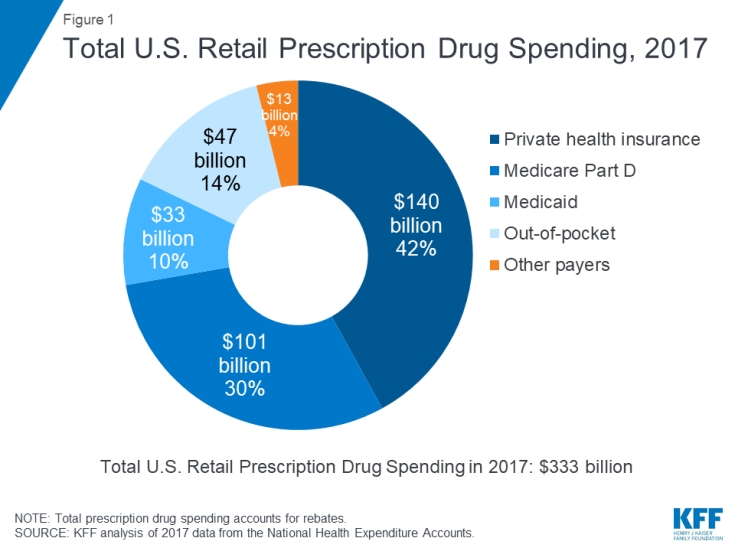
Image from KFF
The IRA also has an “exemption principle” that specifically refers to: drugs that account for less than $200 million in Medicare spending annually; orphan drugs with only one indication; human-derived Whole blood or plasma biological products, etc.
For small biopharmaceutical companies, if a drug under category B or D accounts for more than 80% of its total revenue, it can be “temporarily exempt” in 2026-2028, Not to be negotiated.
Such a price cut,
America’s Innovative Drug Association Hit?
On the eve of the passage of the IRA, the National Association of Manufacturers (NAM), the nation’s largest industrial trade trade organization, had spent a six-figure budget in an advertisement that said “cutting prices will prevent life-saving drugs from entering the market.” According to its statement, for every 10% drop in drug prices, the R&D expenditure invested by enterprises will decrease by 17% accordingly.
“Fears are overblown,” said Loren Adler, director of the Brookings Schaeffer Health Policy Initiative at the University of Southern California. The IRA bill has little impact on new drug development and may even encourage innovation, research and development.
“On the one hand, new drugs can avoid price negotiation, and pharmaceutical companies will be more motivated to introduce new drugs and promote their listing with higher prices to offset the loss of sales after negotiation. On the other hand, Negotiated drugs are those that have been on the market for more than a decade and are already facing generic competition,” she said.
Japan or “Success Model”. In 1988, the country launched a comprehensive centralized procurement involving more than 13,000 drugs. Since then, Japan has adjusted the prices of medical insurance catalogues every two years, with an average drop of more than 6%. The relevant price control measures continue to this day.
This has indeed caused a shock in the industry, and pharmaceutical companies with insufficient competitiveness have successively closed their doors. In 1995, there were 1,512 pharmaceutical companies in Japan. Eight years later, nearly one-third of businesses have disappeared.
During the period, the Japanese government introduced a number of policies to encourage innovation. Around 2000, a number of blockbuster innovative drugs in Japan were listed, the stock prices of related companies rose, and overseas business grew significantly.
The “2021 Global Pharmaceutical Companies TOP 50 List” released by the American “Pharmaceutical Manager” magazine shows that 8 Japanese companies are on the list. One is in the top 10.
In order to better face the opposition of the industry and the doubts of the public, the Congressional Budget Office (CBO) also calculated an account. The results show that after the implementation of price negotiation, about 10 new drugs may be blocked from the market in the next 30 years, accounting for about 1% of the total. During the same period, there were more than 1,000 potential new drugs on the market.
American Drug Wonders:
The longer a drug is on the market, the more expensive it may be
From the previous article, the US version of “Medical Insurance Negotiations” is very focused, that is, exclusive original research drugs, long time on the market, and high medical insurance costs.
The ultimate goal of the negotiation is that the longer the time to market, the greater the price reduction, and the aim is to reduce the medical burden.
U.S. health care spending is “one of the best” among advanced economies, compared to the health outcomes people receive, according to Bill Adams, chief economist at Cofax Bank. Americans pay 2-3 times more for prescription drugs than other countries, according to The Wall Street Journal.
And, more and more expensive.
In June, the Journal of the American Medical Association published a report titled “Trends in Prescription Drug Pricing 2008-2021” by Harvard Medical School researchers. Its content shows that from 2008 to 2021, the median drug listing price in the United States rose from $2,115 to $180,007, an increase of nearly 90 times. During the same period, the median increase in net drug prices was 115.5 times.
“U.S. net prescription drug prices are growing more than three times faster than inflation,” according to another University of Pittsburgh study.
This is puzzling. The United States is the country with the largest output of innovative drugs in the world, and the drug market is extremely rich. But why are drug prices so high?
Answer or a bill for free pricing, patent protection, etc. specific to that country.
In order to recover costs and maintain innovation, the U.S. Free Pricing Act allows U.S. pharmaceutical companies to set their own prices and negotiate prices with commercial insurance companies and pharmacies.
Most commercial insurance companies in the United States entrust it to private companies such as Pharmacy Benefit Management (PBM). The latter acts like an intermediary.
In order to get themselves into the insurance catalog, pharmaceutical companies need to give “benefits” to PBMs and insurance companies, either discounts or rebates.
How much the discount is is an “unspoken secret”. A report shows that in 2015, the sales of prescription drugs in the United States reached 425 billion US dollars, and nearly 30% were returned to PBMs and insurance companies.
If PBM wants more points, and pharmaceutical companies want to maintain high profits, they can only increaseprice.
A survey by the US non-profit organization AAN shows that from 1991 to 2001, the price per milliliter of insulin in the United States rose by an average of 2.9% per year. From 2002 to 2012, the average annual increase was 9.5%. By 2012-2016, it reached 20.7%.
“At the beginning of 2021, the prices of more than 300 drugs in the United States will increase. On the first day of 2020, the prices of 250 drugs will increase, with an average increase of about 5%. In the first 10 days of 2019, 490 drugs will increase in price. The price increase of prescription drugs…” Some analysts said that the price increase of prescription drugs is a “New Year’s gift” from pharmaceutical companies.
This leads to a “spectacle”: the longer the drug has been on the market, the more expensive it can be. After the patent expires, the price may drop as the generic drug is launched.
The reason for saying “or” is that in order to maintain high profits, pharmaceutical companies will continue to enrich the patents of their own products to extend the term of patent protection and prevent generic competition.
I-MAK, a nonprofit organization dedicated to drug patent protection, reports that hundreds of patent applications have been filed for the 12 best-selling drugs in the United States. As a result, the monopoly period of related drugs far exceeds the 20 years stipulated by the US patent law.
The aforementioned Harvard Medical School researcher wrote in the New York Times that in order to deal with the current severe drug price situation, the United States should control the price of drugs when they are on the market and start price negotiations.
However, attempts have been frustrated. Even if the IRA passed this time, it was full of ups and downs. The Wall Street Journal, citing people familiar with the matter, reported that the pharmaceutical industry has been lobbying for a “softer” negotiating package. According to the original version, the federal government has the right to “bargain prices” on up to 250 drugs, regardless of the drug’s age on the market or the degree of innovation.
Ronny Gal, an analyst at investment research firm Bernstein, estimates that after the IRA is enacted, pharma profits will be reduced by 3%-5%. In response to this result, pharmaceutical companies will inevitably consider the pricing of new drugs in the future. This may push up the market price of new drugs in disguise.
The Tax Foundation, a US tax law research organization, also poured cold water: the lack of bipartisan support makes it difficult to guarantee the durability of the IRA Act provisions.
References:
[1]New Reforms to Prescription Drug Pricing in the US Opportunities and Challenges.JAMA.Published online August 19,2022.doi:10.1001/jama.2022.15268
[2]FACT SHEET: The Inflation Reduction Act Supports Workers and Families. The White House
[3]Medicare.gov
[4]How Does Prescription Drug Spending and Use Compare Across Large Employer Plans,Medicare Part D,and Medicaid?.KFF
Source: Medical Think Tank
Editor in charge: Tian Dongliang
Hot text recommendations



* The medical community strives for the accuracy and reliability of its published content at the time of review, but does not care about the timeliness of published content, and the accuracy and completeness of cited material (if any).It does not assume any responsibility for the outdated content, possible inaccuracy or incompleteness of the cited information, etc. Relevant parties are requested to check separately when adopting or using it as a basis for decision-making.