By Nangong Mingyue
Source: stem cellists say
Natural killer cells (NK cells) are a type of innate lymphocytes that have the ability to identify and eliminate virus-infected cells and tumor cells. With the subversive success of CAR-T cell therapy, interest in the potential of NK cells in anti-tumor immunotherapy has doubled.
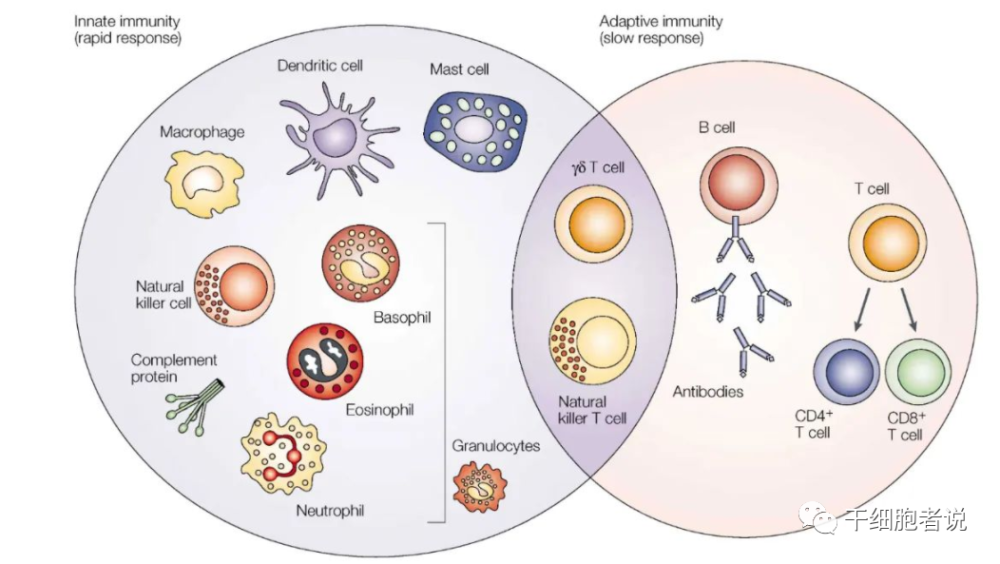
Figure 1 NK cells and other immune cells
In the clinic, the first-generation NK cell therapy has achieved promising results, showing encouraging efficacy and good safety. A review in the top international journal Nature Reviews Cancer(IF=69.800), the author introduces various methods on how to increase the toxicity and lifespan of NK cells, meet the challenges and catch Seize opportunities and guide the design of next-generation NK cell products from lessons learned from clinical development.
–01–
NK cell source and donor selection
NK cells were discovered in the 1970s and are primarily associated with killing infectious microorganisms and malignantly transformed allogeneic and autologous cells. In humans, NK cells are derived from CD34+ co-lymphoid progenitor cells. It is estimated that the half-life of NK cells is about 7-10 days, and they play a key immunoregulatory function by interacting with DC cells.
NK cells come from a wide range of sources, including peripheral blood, umbilical cord blood, pluripotent stem cells (ESC and iPSC) and NK cell lines (such as NK92 cell line, etc.). All sources can provide clinically meaningful cell doses suitable for the preparation of CAR-NKs and have transitioned to human studies. Each source has advantages and potential challenges. CAR-NK cells have been successfully engineered from different platforms including cord blood.
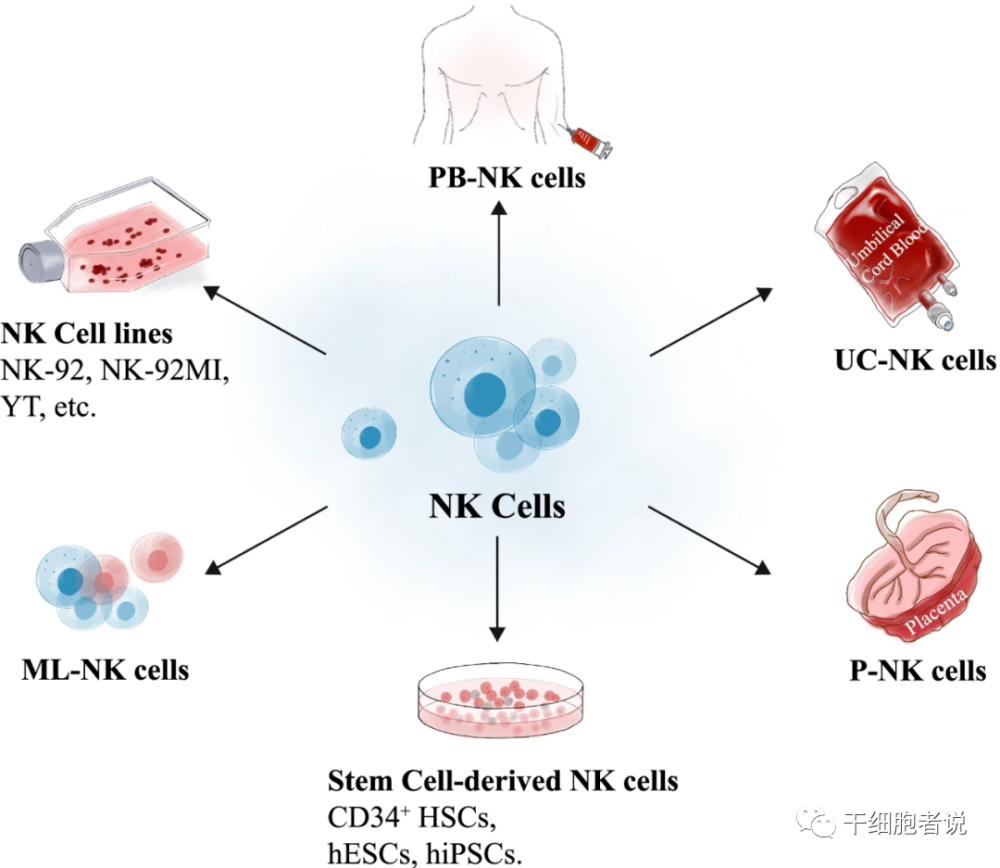
Figure 2 Source and donor selection of NK cells
▉Peripheral or cord blood source
Primary NK cells can be harvested from peripheral blood (PB-NK cells) or from umbilical cord blood (CB-NK cells). PB-NK cells, a platform for the first successful delivery of CAR constructs into NK cells led by Dario Campana in 2005. PB-NK cells provide the basic material for various products currently in clinical trials.
▉Source of induced pluripotent stem cells
iPSCs are a source of NK cells. iPSCs can be efficiently expanded and differentiated in vitro. A large number of NK cell products with the same genetic background can be produced by iPSC. The preparation of iPSC needs less seed cells, can be cultured in large quantities, has low cost, can be self-supplied, and has low immunogenicity.
iPSC-derived NK cells have two disadvantages. First, they express low levels of CD16. Second, iPSCs may contain “epigenetic memory” that may influence the development of specific cell lineages distinct from donor cells.
▉NK cell line source
NK-92, the first NK cell to receive FDA approval for clinical trials, is a homogeneous, immortalized NK lymphoma cell line that can be expanded ex vivo. NK-92 cells lack the expression of most KIRs and are therefore unlikely to be repressed.
However, NK-92-derived cell products need to be irradiated prior to patient administration, which affects long-term persistence and overall therapeutic potential in vivo. Another disadvantage is that NK-92 cells lack the ability to mediate cell killing via ADCC due to lack of CD16 expression.
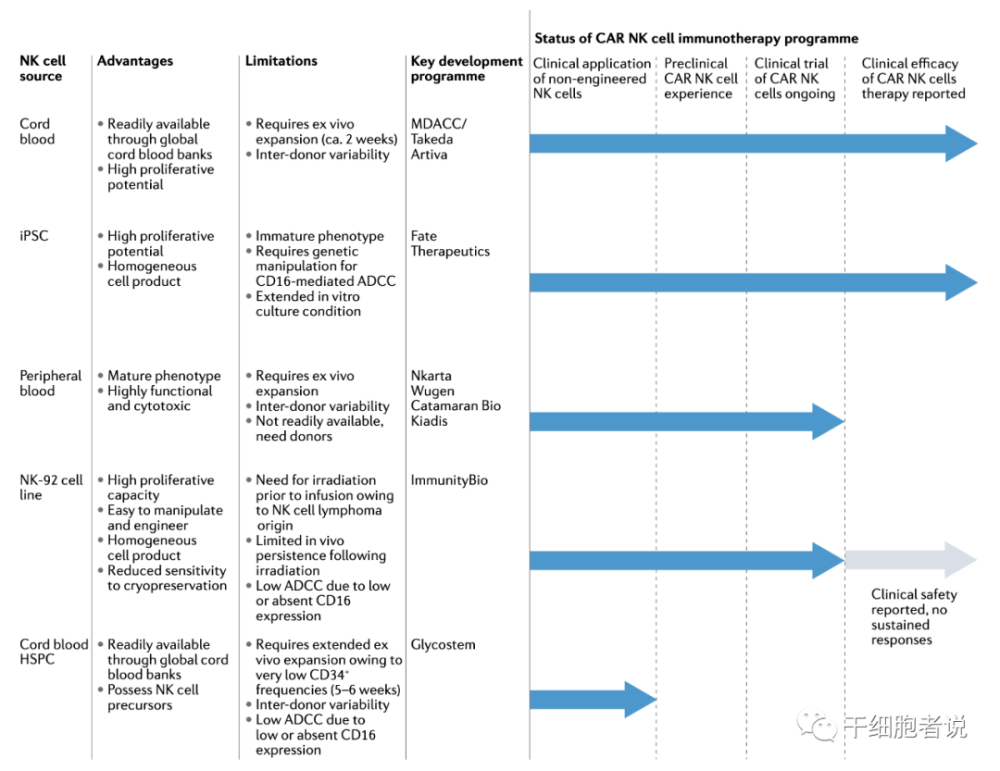
Figure 3 Advantages and limitations of NK cells from different sources
–02–
How to enhance NK cell function
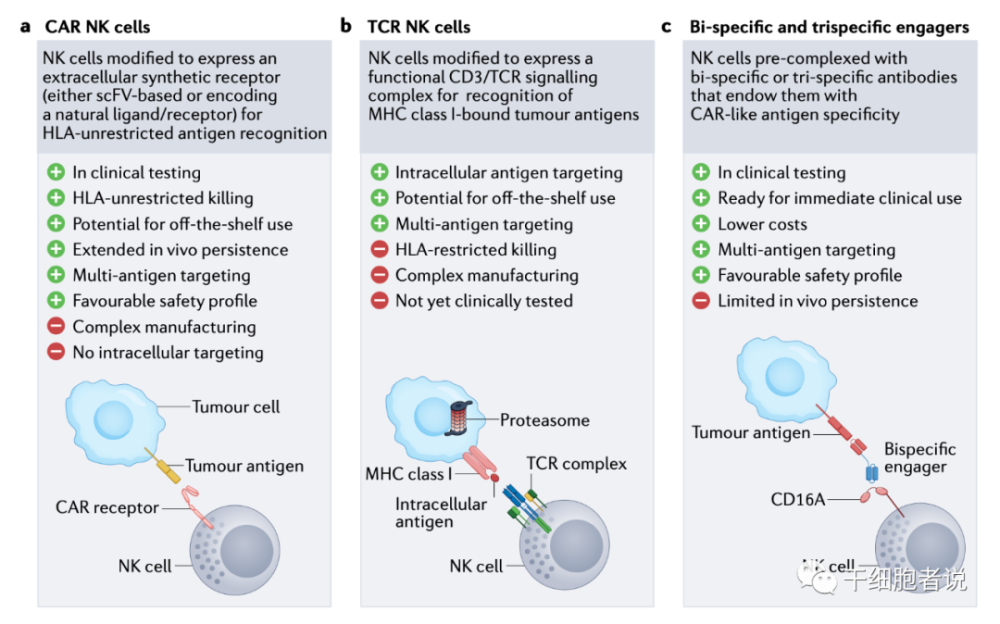
Figure 3 Strategies to enhance NK cell specificity
▉Chimeric Antigen Receptor
CARs are synthetic fusion proteins that include an extracellular antigen recognition domain and an intracellular signaling moiety that triggers cellular activation. CARs can be expressed on immune effector cells to reprogram their specificity for specific targets. CAR-T cell therapy was the first to emerge. Since then, CARs traditionally designed for T cells have been used to generate CAR-NK cells, which studies have shown can target specific tumors while maintaining a desirable safety profile. Intracellular antigens exist in the form of peptide-HLA complexes, which are detected by TCR. Engineered NK cells to express TCR can detect this peptide. TCR-NK cells, which have been shown to elicit anti-tumor responses.
▉NK Cell Adapter
NK cell adapters, which direct NK cells to the tumor site, trigger a strong anti-tumor response by triggering activating receptors on NK cells, while binding to target antigens on tumor cells. Development strategies include three-specific or four-specific designs, targeting multiple antigens on tumors, or by cross-linking cytokine moieties, promoting NK expansion and survival, aiming to enhance anti-tumor effects. The use of cell adapters thus represents a simpler and lower cost manufacturing process to provide products capable of inducing CAR-like activity.
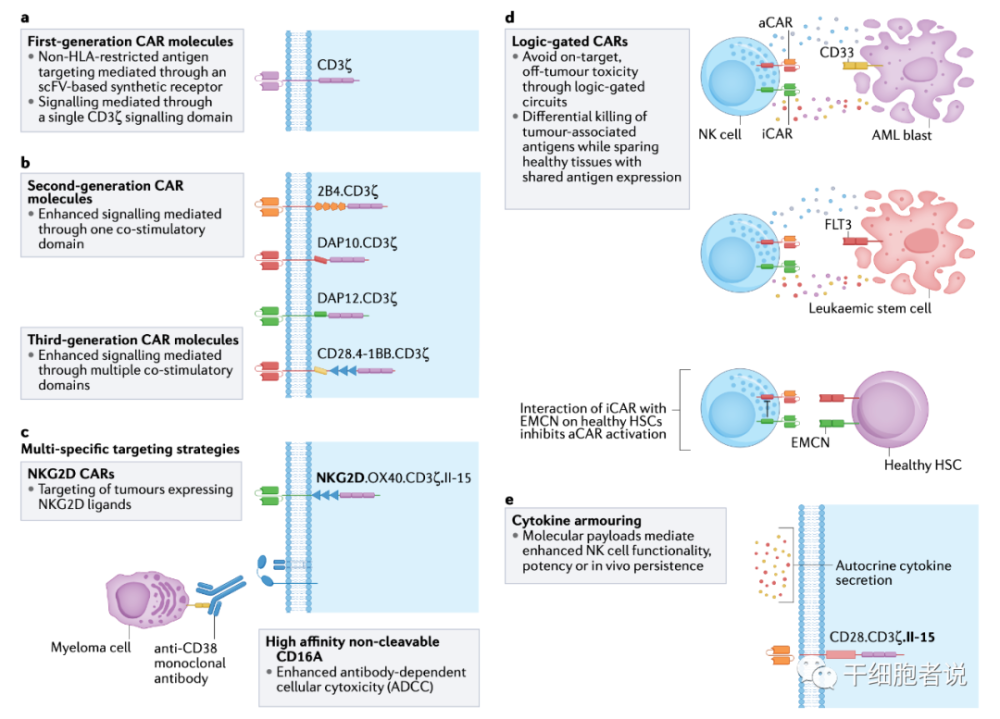
Figure 4 Principles and strategies of CAR design
▉Cytokines
Cytokines can enhance NK cytotoxicity and promote NK cell proliferation. Continuous ex vivo stimulation, NK cells are “cytokine-addicted” and reduce persistence when these cells are infused without in vivo cytokine support. Through genetic engineering, NK cells are modified to produce cytokines that maintain cellular potency, proliferation, and persistence. This autocrine maintenance is of great interest and CAR-NK cells are emerging.
▉Overcoming immunosuppression
Tumors often exhibit abnormal metabolic behavior, resulting in elevated levels of lactate and increased concentrations of toxic catabolites, adenosine and reactive oxygen species, in the tumor-surviving environment. In addition, within the tumor microenvironment (TME), there are proliferating and dysfunctional vasculature and immunosuppressive cell subsets, rendering the various immune effector cells entering the TME ineffective.
To overcome immunosuppression, current strategies focus on two areas:
Altering the metabolic makeup of tumors or modifying gene expression programs in immune cells to protect them from inhibitory metabolites in the TME;
Blocking CD73 on tumor cells by small molecule inhibitors or antagonist antibodies overcomes adenosine-mediated immunosuppression.
In preclinical studies, gene editing to delete adenosine A2A receptors in CAR-T cells and CAR-NK cells has shown anti-tumor efficacy.
▉BlockingImmune Checkpoints
Tumors can evade immune surveillance (eg, the involvement of immune checkpoints), and NK dysfunction can be reversed with the use of corresponding mAbs. However, modulation of patients’ immune cells by monoclonal antibodies requires multiple infusions. With the advancement of gene editing technology, NK cells can be stably modified to enhance NK cell function. Furthermore, after identifying CISH (cytokine-induced SH2-containing protein) as a key negative regulator of NK cell function, CAR-NK cells lacking this intracellular cytokine checkpoint were engineered for metabolic fitness and antitumor activity Significantly improved.
▉Enhanced NK cell trafficking to tumors
The ability of NK cells to enter and penetrate tumors is the key to effective anti-tumor immunity. Similar to other immune cells, NK cells are directed to tumor sites through the dynamic interaction of chemokine receptors and their cognate ligands secreted in the TME. Due to the rapid loss of chemokine expression due to internalization and degradation, genetic engineering to equip NK cells with stable ectopic chemokine receptors is a direction for future CAR-NK development.
–03–
Clinical Experience Summary
CAR-T cell therapy has allowed some patients to achieve effective treatment for up to ten years. NK cells have unique antitumor effects, MHC-independent cytotoxicitystrong>, production of cytokines and immune memory and other functions, making it a key role in the innate and adaptive immune response system. Early clinical data show that NK cells are very suitable for use in an allogeneic therapeutic setting. Although the safety results are encouraging, further studies are needed to elucidate whether allogeneic NK cells are able to escape T cell rejection for long-term persistence.
Clinical studies have shown that NK cells are safe and effective in the treatment of hematological malignancies. Importantly, regardless of the approach, NK cell therapy has consistently shown a favorable safety profile. To date, no CRS or GVHD have been observed. However, most reported successes are limited to hematological malignancies, not all patients respond to NK cell therapy, and some eventually relapse.
In any case, CAR-NK cell therapy is a promising area of clinical research with good safety and preliminary efficacy in certain cancer patients. It is believed that CAR-NK cell therapy may lead to a revolutionary progress in tumor immunotherapy.

Rezvani
Rezvani, Professor, Division of Cancer Medicine, The University of Texas MD Anderson Cancer Center. His laboratory has focused extensively on the protective role of natural killer (NK) cells in hematological malignancies and solid tumors, as well as enhanced strategies to target various cancer-killing functions. Research areas include immunotherapy, immunology, genetics, CAR T cells, CAR-NK cells.
Current Rezvani lab discoveries and research topics include: comprehensive analysis of NK cells and their receptors in cancer after hematopoietic stem cell transplantation using mass spectrometry (CyTOF) and transcriptome analysis; designing NK cells cells to express chimeric antigen receptor (CAR) and cytokine genes to enhance their effector function and persistence; understand mechanisms of NK immune evasion and use gene editing tools to target checkpoints to enhance NK effector function; utilize CARs and Gene editing tools such as CRISPR enhance the function of T cells against viruses and cancer antigens.
References:
[1] Laskowski, T.J., Biederstädt, A. & Rezvani, K. Natural killer cells in antitumour adoptive cell immunotherapy.Nat Rev Cancer(2022).< /p>
[2] Maskalenko et al., Harnessing natural killer cells for cancer immunotherapy: dispatching the first responders. Nature Reviews Drug Discovery, (2022).
[3] Le Saux and Schvartzman Advanced Materials and Devices for the Regulation and Study of NK Cells.Int. J. Mol. Sci., (2019).
[4] Zhang et al.,. CAR-NK cells for cancer immunotherapy: from bench to bedside.Biomarker Research,(2022)