On July 25, the State Food and Drug Administration approved the registration application for the addition of azvudine tablets for the treatment of new coronavirus pneumonia by Henan True Biotechnology Co., Ltd. with conditions.

Screenshot of the official website of the State Food and Drug Administration
And on July 20, 2021, the State Food and Drug Administration has conditionally approved Azvudine in combination with other reverse transcriptase inhibitors for the treatment of HIV-1 infection in adults with high viral load patient.
With the conditional approval of the new indication, Azvudine can be used to treat adult patients with common new coronary pneumonia. The State Food and Drug Administration requires the marketing authorization holder to continue to carry out relevant research work, complete the conditional requirements within a time limit, and submit the follow-up research results in a timely manner.
Azvudine is an oral dual-target nucleoside antiviral drug with significant and broad-spectrum antiviral effects against HIV, HCV, EV71 and HBV in vitro.
The new coronavirus is a typical enveloped single-stranded positive RNA virus, and RNA synthesis requires nucleosides and nucleotides as raw materials. Nucleoside antiviral drugs can enter host cells by imitating natural nucleosides, and then converted into nucleoside triphosphates through the catalytic action of kinases, and embedded in viral RNA during viral RNA synthesis, resulting in the termination of viral RNA chain synthesis. to inhibit virus replication.
Azvudine previously demonstrated promising efficacy and safety in Phase II clinical trials for the treatment of HIV infection.
In July 2020, the Azvudine R&D team—Chang Junbiao’s team at Henan Normal University—published an article online in Advanced Science on the treatment of mild and common COVID-19 patients with Azvudine randomized controlled clinical trials.

Paper screenshot
The study included 20 patients with mild and normal COVID-19, who were randomly assigned to receive azvudine and symptomatic treatment or standard antiviral and symptomatic treatment. The results showed that the mean time to first visit was 2.6 days in the azvudine-treated group, which was shorter than 5.6 days in the control group; no adverse events occurred in the azvudine-treated group, and 3 adverse events were reported in the control group.
Preliminary results suggest that azvudine may shorten the time to negative nucleic acid in patients with mild and normal COVID-19 compared with standard antiviral therapy, according to the study. Therefore, it is necessary to conduct a clinical trial of azvudine in the treatment of new crown with a larger sample size.
On December 6, 2021, Chang Junbiao’s team republished the research results of Azvudine in the treatment of new crown in the Nature sub-journal Signal transduction and targeted therapy.

Paper screenshot
In Covid-19-infected rhesus macaques, oral azvudine treatment reduced viral load, reduced inflammation and organ damage, and reduced ground-glass opacities on chest X-rays. At the same time, a randomized single-arm clinical trial of compassionate use showed that oral administration of azvudine cured all 31 patients with COVID-19, the time to negative nucleic acid was 3.29 ± 2.22 days, and 5 patients developed mild and transient dizziness, nausea.
According to ClinicalTrials.gov, there are three registration studies of Azvudine for the treatment of COVID-19, none of which are currently published.
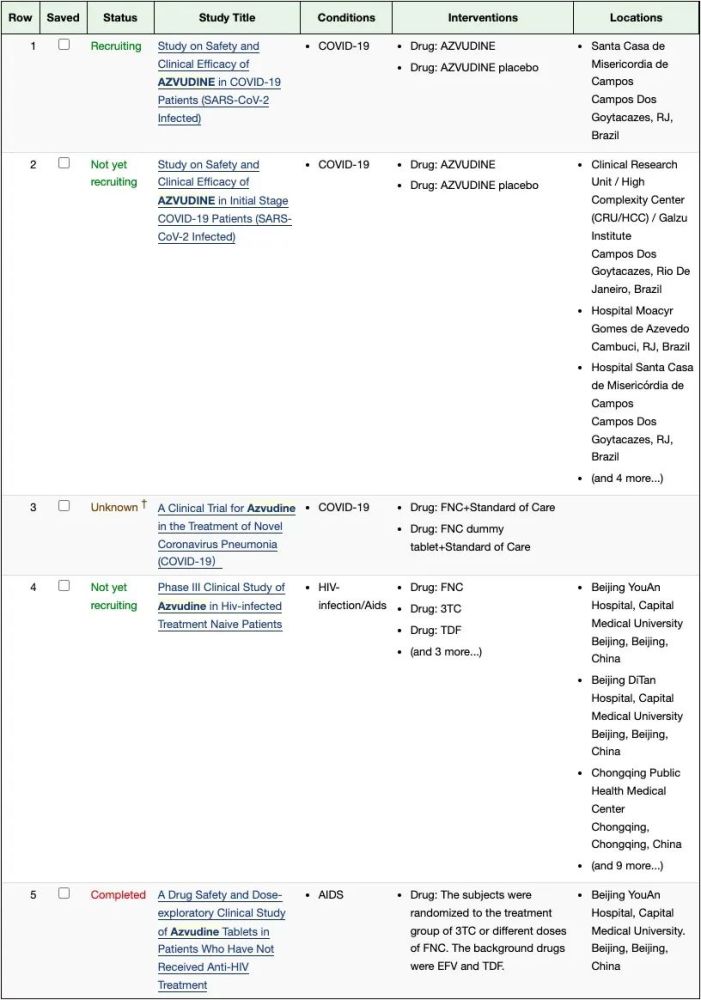
Screenshot of ClinicalTrials.gov
On July 15, 2022, the real biotechnology WeChat official account issued a tweet announcing that the results of the Phase III clinical trial of Azvudine Tablets for the treatment of new coronavirus pneumonia indications have reached expectations.

Tweet screenshot
According to the tweet, the Phase III clinical trial of Azvudine adopts a multi-center, randomized, double-blind, placebo-controlled clinical trial design. The clinical trial results show:
1. Significantly improve clinical symptoms: Azvudine Tablets can significantly shorten the symptom improvement time in patients with moderate novel coronavirus infection pneumonia, and improve the clinical symptoms improvement. The proportion of patients to achieve clinically superior results. The proportion of subjects whose clinical symptoms improved on the 7th day after the first dose was 40.43% in the azvudine group and 10.87% in the placebo group (P value
2. Inhibition of the new coronavirus: Azvudine has the activity of inhibiting the new coronavirus, and the virus clearance time is about 5 days.
3. Safety: Azvudine tablets are generally well tolerated, and the incidence of adverse events is not statistically significant between the Azvudine group and the placebo group differences, with no increased subject risk.
However, no research papers have been published on the Phase III clinical trial of Azvudine. The State Food and Drug Administration also stated in the notice that it requires marketing license holders to continue to carry out relevant research work, complete the conditional requirements within a time limit, and submit follow-up research results in a timely manner. (Planner: z_popeye|Producer: gyouza)
The source of the title map: Screenshot of the official website of the State Food and Drug Administration