▎WuXi AppTec Content Team Editor
Viruses are often considered so simple that they cannot even survive without the help of the cells of other organisms. However, these little viruses, unable to survive on their own, have developed a superb ability to make full use of their environment for their own benefit.
A new study published in Frontiers in Microbiology has just found that They are actually able to “monitor” the state of the host cell to decide when it’s better to sit still and when it’s better to replicate frantically and break out of the cell in order to infect more bacteria.

Bacteriophages studied by scientists at the University of Maryland, Baltimore County (UMBC) specifically infect a type of bacteria that grow fimbriae, or flagella.
Bacteria of the genus Caulobacterales, for example, were first observed by researchers in two distinct forms, one with elongated fimbriae, or flagella, and Free-swimming; the other form has no fimbriae or flagella, in which case they are attached to surfaces. The bacteria of the genus Cobacteria usually live in nutrient-poor environments and are widely distributed, but when they find a good place to grow, they change into the latter form and begin to proliferate, producing large populations of cells, the researchers said.
And the phages that infect this bacteria seem to be very good at timing, taking advantage of the bacteria’s bloom and rushing out of the cell to infect as many bacteria as possible around them.
So, how do phages master their timing? The researchers found that bacterial cells express a protein called CtrA, which controls when cells produce fimbriae, or flagella, and that levels of the protein fluctuate up and down during the cell cycle. Bacteriophage’s genome has a large region that can bind to CtrA protein. These binding sites mean that phage can monitor the level of CtrA in bacterial cells.
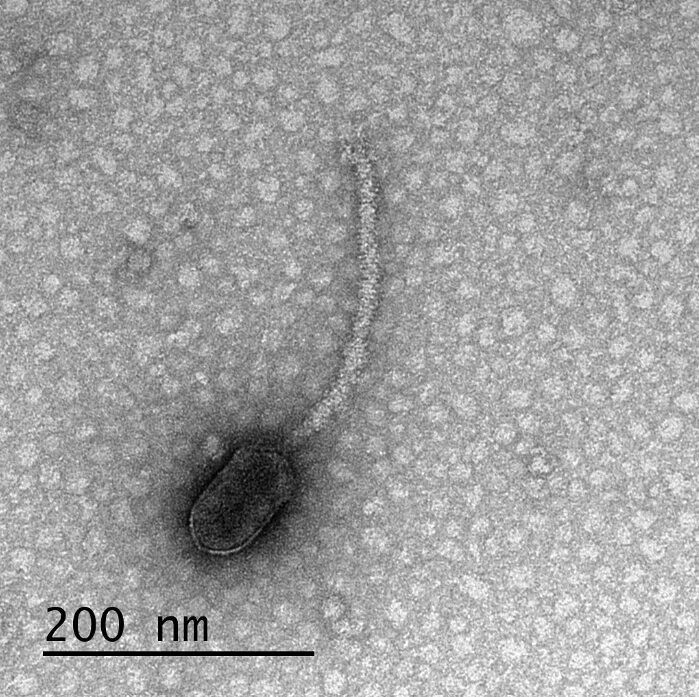
▲For the first time, this phage virus was found to have a binding site for CtrA, thereby monitoring the expression of CtrA in host bacteria to “decide” whether To replicate in large numbers and leave the cell (Image credit: Reference [2]; Credit: Tagide deCarvalho/UMBC)
What’s more, this surveillance ability of viruses is not just the unique ability of a certain phage. Through detailed genome analysis, the research team found that many different types of phages have CtrA protein binding sites, and the bacterial hosts infected by these phages also grow fimbriae or flagella. Obviously, this is no coincidence.
Study leader Professor Ivan Erill believes that it is likely that different phages that infect different bacteria have developed the ability to monitor CtrA levels several times during their evolution, this “convergent evolution” , which also shows that this survival skill mastered by the virus is very effective. Therefore, the researchers further speculate that in addition to bacteria-infecting viruses, infection of other organisms cannot be ruled out — even human viruses have similar “surveillance” capabilities.
“Everything we know about bacteriophages, and every evolutionary strategy they’ve developed, has been shown to translate into viruses that infect plants and animals,” added Professor Erill. . “It’s almost taken for granted. So if bacteriophages are spying on their hosts, viruses affecting humans must be able to do the same.” For example, in order to optimize their survival and replication strategies, animal viruses might want to know The type of tissue it is in, or how strong the host’s immune response to infection is.
While the virus may sound more terrifying, “knowing yourself and knowing your enemy is a good way to fight,” the researchers say, and these new discoveries could lead to new treatments. “If we’re going to develop an antiviral drug, and you know the virus is monitoring a certain signal, then maybe we’ll be able to trick the virus.“
References:
[1] New research finds that viruses may have ‘eyes and ears’ on us. Retrieved Sep. 26, 2022 from https:https://phys.org/news/2022-09 -viruses-eyes-ears.html
[2] Elia Mascolo et al, The transcriptional regulator CtrA controls gene expression in Alphaproteobacteria phages: Evidence for a lytic deferment pathway, Frontiers in Microbiology (2022). DOI: 10.3389/fmicb.2022.918015