On August 22, Gilead Sciences announced that the European Commission (EC) has granted marketing authorization for Sunlenca (lenacapavir) injections and tablets for use in combination with other anti-inflammatory drugs Retroviral drug therapy in multidrug-resistant HIV-infected adult patients. Lenacapavir is a “first-in-class” capsid inhibitor with a multi-stage mechanism of action and has no known cross-resistance to other existing drug classes. HIV patients who show effective responses to current therapies offer a new, 6-monthly treatment option.
Lenacapavir’s Marketing Authorization Application (MAA) is supported by data from the Phase 2/3 CAPELLA trial evaluating lenacapavir in combination with an optimized background antiviral regimen in heavily treated patients Use in multidrug-resistant HIV patients. In the patient population with significant unmet medical need, 83% (n=30/36) of subjects receiving lenacapavir on top of the optimized background regimen achieved an undetectable viral load at Week 52 . In addition, CAPELLA subjects had an average increase in CD4-positive cell counts of 83 cells/µL. The New England Journal of Medicine has published the primary results of the CAPELLA study.
“Lenacapavir helps fill a critical unmet need in patients with complex prior treatment histories and provides a long-awaited twice-yearly option.” University of Paris-Sité “In the CAPELLA study, lenacapavir, in combination with other antiretroviral therapies, demonstrated sustained virological suppression in patients with multidrug-resistant HIV,” said Dr. and clinically meaningful recovery of CD4-positive T cells. Lenacapavir offers an innovative long-acting HIV therapy option that has the potential to change the clinical landscape.”
“After more than three decades of advancing advances in HIV treatment and prevention, Gilead scientists have now delivered an innovative new option for long-acting care,” said Gilead Sciences Chairman and CEO Mr. Daniel O’Day, Chief Executive Officer, said, “lenacapavir is a unique and potent drug with the potential for flexible dosing options. With today’s approval, it will be the first for those battling multidrug-resistant HIV. This is a twice-yearly therapy. Our goal is to provide a variety of long-acting options in the future, which we believe will make a fundamental difference in ending the HIV epidemic.”
Despite significant advances in antiretroviral therapy, there are still many critical and urgent unmet needs for people living with HIV, with treatment-heavy and limited treatment options , and especially in HIV patients who are unable to maintain virological suppression due to the challenges of drug resistance or adherence to complex treatment regimens.
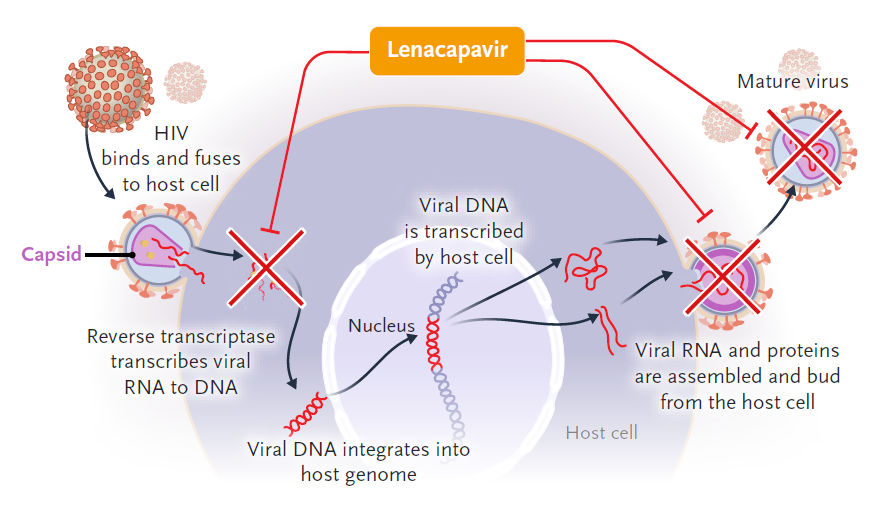
▲The drug mechanism of Lenacapavir against HIV (Image source: Reference [2])
Most antiviral drugs act on only one stage of viral replication, but lenacapavir is designed to inhibit HIV at multiple stages of its life cycle and has There is no known cross-resistance to other drug classes available. Lenacapavir binds to the capsid protein monomer, making the resulting capsid a malfunctioning “substandard product”. It is worth mentioning that in 2021, lenacapavir was included in the Annual Small Molecule List selected by industry drug developers published by the drughunter website—and ranked first on the list. bit.
Lenacapavir is indicated in the European Union for the treatment of adults with multidrug-resistant HIV infection in combination with other antiretroviral drugs. Lenacapavir tablets are approved for oral administration prior to the administration of long-acting lenacapavir injection. Marketing authorization applies to all 27 EU member states, as well as Norway, Iceland and Liechtenstein. European marketing authorization is the latest milestone in the review of lenacapavir by major global regulators. In July this year, the US FDA accepted the resubmission of the New Drug Application (NDA) review materials for lenacapavir with a PDUFA date of December 27, 2022.