In 2013, cancer immunotherapy was named one of the “Top Ten Scientific Breakthroughs” by Science. The success of immunotherapy not only revolutionized the effect of cancer treatment, but also changed the concept of treating cancer. Long-standing attempts to activate a patient’s own immune system to treat cancer have finally been successful. Indeed, the research and development of immune checkpoint inhibitors such as PD-1/PD-L1 pathway and CTLA4 pathway has brought new treatment options for cancer patients. From the back-line to the first-line, to perioperative adjuvant and neoadjuvant, immunotherapy can be said to have penetrated into all stages of tumor management.
In terms of efficacy, immunotherapy is characterized by a “tail effect”. Once the drug has an effect on the tumor, the patient may have a lasting clinical benefit. In terms of safety, the overall adverse reactions are controllable, and most immune-related adverse events (irAEs) occur within 3 months of starting treatment, and are easy to be concerned about.
However, with the extension of clinical trial follow-up time and the increase of real-world research experience, more and more scholars have found that there are delayed and chronic long-term adverse effects in immunotherapy. event. These toxic reactions take a long time from the start of immunotherapy, and once they appear, people are caught off guard, and we have to pay attention. An article recently published in the journal Nature Reviews Clinical Oncology sheds light on this important question.
Delayed immune-related adverse events
Delayed immune-related adverse events were defined as “adverse events that occurred one year after initiation of immunotherapy”. The most common of these adverse events include immune colitis, rash, pneumonia and so on. More than 70% of the patients were still receiving immunotherapy when these adverse events occurred; for others, it occurred after the completion of immunotherapy. This suggests that the timing of immune-related toxicity is less certain and may occur during treatment or after treatment has ended. Therefore, follow-up monitoring of patients during and after treatment is very important.
After immunotherapy, some patients will experience immune-related adverse events lasting more than 12 weeks, which we call “chronic immune-related adverse events”. Such adverse events are not uncommon, and they are easily overlooked due to the long time since initial treatment, and are of particular concern.
Chronic immune-related adverse events can affect many systems in the body
Chronic immune-related adverse events have potential toxicity to various systems of the body, the most common of which are endocrine toxicity and rheumatic toxicity (Figure 1).
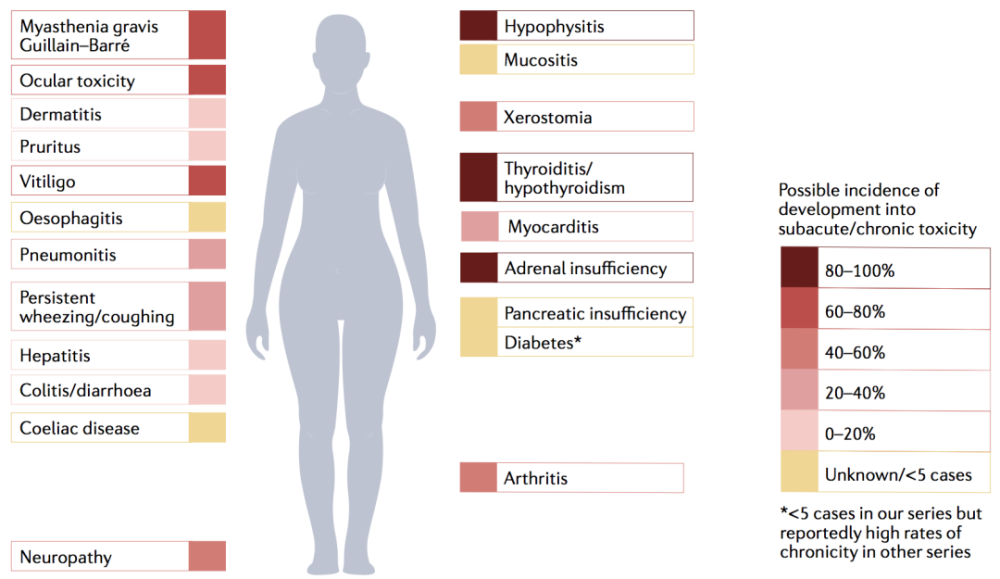 Figure 1. Patterns of chronic immune-related adverse events caused by immune checkpoint inhibitors< /i>
Figure 1. Patterns of chronic immune-related adverse events caused by immune checkpoint inhibitors< /i>
Endocrine toxicity
This type of toxicity occurs in 15%-40% of patients, the more common types being hypothyroidism, hypophysitis, and type 1 diabetes. Hypothyroidism usually occurs about six weeks after receiving immunotherapy. About half of the patients will have transient symptoms of hyperthyroidism due to thyroid destruction and increased thyroid hormone before hypothyroidism occurs. Immune hypophysitis was more common with ipilimumab-containing regimens and persisted once it occurred.
After such toxicities occur, replacement therapy is the mainstay. If hypothyroidism occurs, thyroxine should be taken orally for a long time. Such toxicities often do not require immediate discontinuation of immune checkpoint inhibitor therapy or high-dose corticosteroids.
Rheumatic toxicity
Rheumatic toxicity is related to immune strength. Once the effect of immunotherapy is too strong, in addition to anti-tumor, it may also produce a reaction against the body’s own normal organs, which is rheumatic toxicity. The more common ones are rheumatoid arthritis, polymyalgia rheumatica, polymyositis, and Sjögren’s syndrome. About half of the patients receiving immunotherapy will experience arthritis symptoms for six months to a year after finishing treatment, and some of these patients may have permanent symptoms.
Hormonal and disease-modifying antirheumatic drugs are treatments for these immune-related adverse events. This type of toxicity will have a certain impact on the quality of life of patients, but it is often not serious, and high-dose corticosteroids are generally not required.
Gastrointestinal toxicity
Immune colitis is a common adverse event, mainly including diarrhea, abdominal pain, blood in the stool and other symptoms. When these symptoms occur, pay attention. In addition, when immune-related hepatitis occurs, non-specific symptoms such as discomfort and myalgia often occur, and liver function should be followed up. Physicians generally decide whether to recommend hormonal intervention based on the severity of the adverse event.
Pulmonary toxicity
Immune pneumonitis is a common immune-related adverse event with anti-PD-1 therapy, sometimes resulting in death. After receiving immunotherapy, dry cough, decreased blood oxygen saturation, interstitial changes in lung imaging, etc., should be paid special attention to exclude immune pneumonia. Doctors generally decide whether to receive hormone therapy based on the severity of the adverse event.
Other toxicity
Immune-related adverse events may also occur in other organs, including cardiovascular toxicity (immune myocarditis, acute vasculitis, etc.), skin toxicity (rash, pruritus, vitiligo), neurotoxicity, hematology toxicity, etc. During and after treatment, close follow-up and attention should be given.
Chronic immune-related adverse events can be fatal and rare but should not be ignored
Fatal adverse events are rare but cannot be ignored. After immunotherapy, adverse events can be fatal, and even patients with durable efficacy benefits and older populations are more likely to have potentially fatal adverse events. The most common include myocarditis, pneumonia, hepatitis, colitis and so on.
Summary and Outlook
Individualized whole-process management of tumors is well reflected in the follow-up of immune-related adverse events. These adverse events most frequently occur within 3 months of starting treatment, so safety should be monitored more closely at the start of treatment. During and after immunotherapy, related adverse events will also occur, and regular follow-up is still necessary.
For patients who continue to benefit from treatment, even if no serious adverse events occur during immunotherapy, follow-up should be standardized after treatment, because immune-related adverse events can occur after treatment. Occurs in a chronic and more severe state.
In addition, since immunotherapy may have effects or interactions with other immune processes, if there are changes in other disease symptoms, the relationship to immunotherapy should also be considered (figure 2). Monitoring and controlling immune-related adverse events will provide the most important safety guarantee for the long-lasting efficacy of immunotherapy.
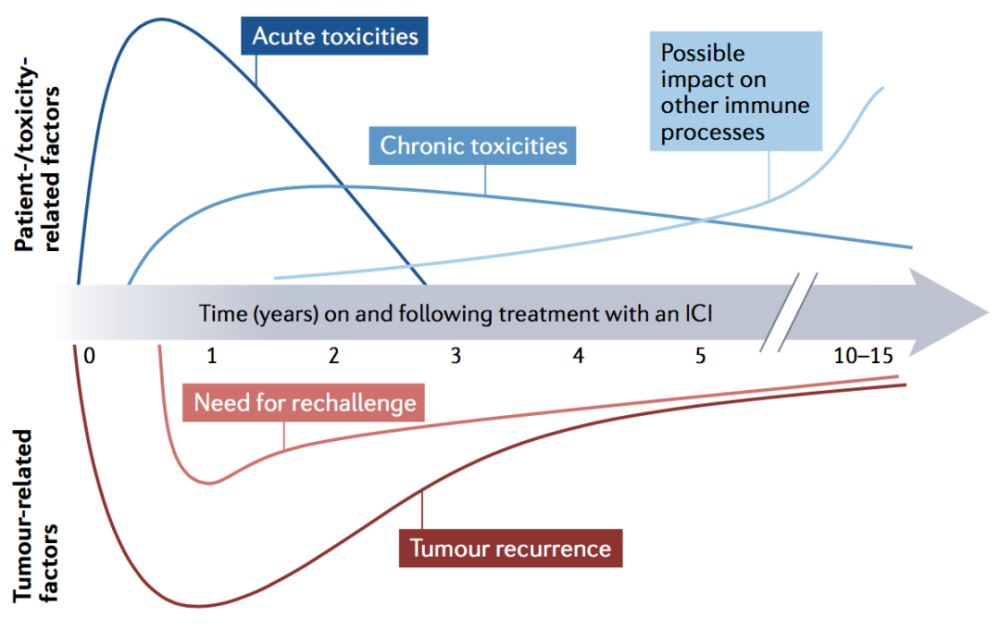 Figure 2. Important issues to be concerned about at different stages of immunotherapy
Figure 2. Important issues to be concerned about at different stages of immunotherapy