▎WuXi AppTec Content Team Editor
CAR-T therapy has shown amazing efficacy in the treatment of blood cancers, and Emily Whitehead, the first pediatric patient to receive CAR-T therapy, has
10 years cancer free
. It gives blood cancer patients the hope of a cure. For many patients, however, cancer still returns. Recently, two research teams from Harvard University and Stanford University published an article in the journal Nature Medicine, each independently revealing that regulatory T cells mixed in the manufacturing process of CAR-T therapy may be responsible for the ineffectiveness of CAR-T therapy. One of the reasons for the best. This discovery is expected to further improve the way CAR-T therapy is manufactured and improve the efficacy of future therapies.
The manufacturing process of CAR-T therapy requires obtaining T cells from patients, genetic engineering in vitro, and expressing chimeric antigen receptors (CARs) targeting tumor antigens on T cells. On the surface of the CAR, these CARs will guide T cells to the vicinity of tumor cells expressing the antigen, and activate T cells to kill tumor cells.
T cells obtained from patients include many types, such as CD8-positive cytotoxic T cells, and CD4-positive helper T cells. The researchers compared the CAR-T cell types in cancer patients who achieved remission and those whose cancer had recurred, hoping to find clues about the emergence of drug resistance.
Using technologies such as single-cell RNA sequencing, the two research teams assessed the types of CAR-T cells in patients after receiving CAR-T therapy. Surprisingly, they all found that there is a small fraction of regulatory T cells (Treg) in CAR-T cells. This caught the researchers’ attention.
Regulatory T cells have the opposite function to effector T cells in the immune system. Usually, effector T cells differentiate into cytotoxic T cells after being activated by antigens presented by antigen presenting cells (APCs). cells that, after recognizing antigens on the surface of target cells, perform cytotoxic functions and destroy the target cells.
Regulatory T cells can be activated in a variety of ways to suppress surrounding effector T cells. Includes the expression of anti-inflammatory cytokines such as IL-10, TGFβ and IL-35; and the depletion of cytokine IL-2 that activates effector T cells. They can also indirectly inhibit the activation of effector T cells by interacting with APCs. An important feature of the immunosuppressive ability of regulatory T cells is a phenomenon known as “bystander suppression”. It means that although a regulatory T cell is activated by a specific antigen, after activation, it can inhibit the activity of effector T cells targeting other antigens. These characteristics allow regulatory T cells to become
Treatment of autoimmune and inflammatory diseases
The ideal tool for
.
However, finding regulatory T cells in CAR-T therapy is not a good thing. These regulatory T cells may also be activated by tumor-expressed antigens, thereby inhibiting surrounding cells. The activity of toxic CAR-T cells reduces the efficacy of CAR-T therapy.
Harvard teams have found that whether a patient responds is related to the regulatory T cell component of CAR-T cell therapy. “In responders, the proportion of regulatory T cells averaged 2%, while in non-responders, the proportion increased by 5-6%.” Harvard University study lead author, Harvard Biotechnology said Nicholas J. Haradhvala, a doctoral student in the Department of Physics.
To test whether regulatory T cells reduce the effect of CAR-T therapy, the researchers tested in a mouse model mixing CAR-carrying regulatory T cells (CAR-Treg) into Results in CAR-T cell therapy. They found that if the proportion of CAR-Treg reaches 5% of CAR-T cell therapy, it can significantly increase tumor proliferation in mice, while other CD4-positive T cells mixed with non-CAR-Treg will not have this effect.
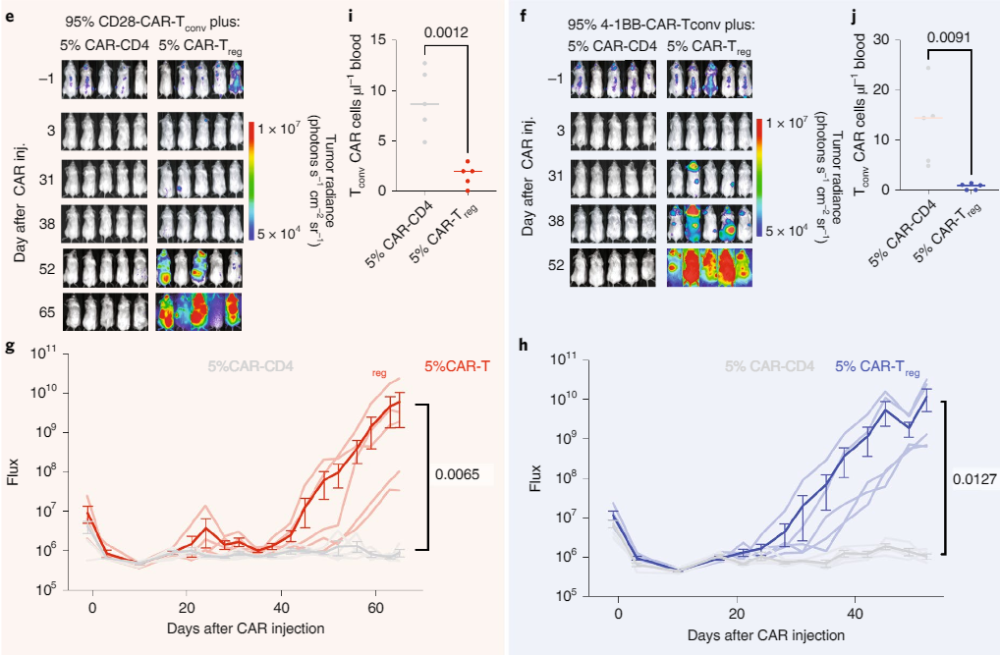
▲Adding CAR-Treg to CAR-T cell therapy significantly reduces the therapeutic effect (Image source: Reference [2])
In the Stanford team’s study, the researchers also found that the proportion of CAR-Treg cells correlated with cancer progression and the neurotoxicity of CAR-T cell therapy. The higher the proportion of CAR-Treg cells, the higher the risk of disease progression and the milder the symptoms of neurotoxicity.
Since this result was independently observed by research teams at Harvard and Stanford, it means that CAR-Treg cells may indeed be It will affect the efficacy of CAR-T therapy. “Two different research teams used different methods to reach the same conclusion, which gives you more confidence that CAR-Treg is working,” he said.
This also brings hope. Dr. Neelapu said the reasons why CAR-T therapy does not work can be grouped into three main categories: differences in individual patient biology, issues related to tumor biology, and issues related to CAR-T manufacturing. This discovery sheds light on problems associated with CAR-T manufacturing that are relatively easier to solve. If regulatory T cells can be eliminated in the CAR-T manufacturing process, it is possible to further improve the effect of CAR-T therapy.
It is expected that this research will lead to a more efficient CAR-T therapy for the majority ofbenefit cancer patients.
As WuXi AppTec’s CTDMO focused on cell and gene therapy, WuXi Bioscience is committed to accelerating and transforming the development, testing, manufacturing and commercialization of gene and cell therapy and other high-end treatments. WuXiXi can help global customers bring more innovative therapies to the market as soon as possible for the benefit of patients. If you have relevant business needs, please click the picture below to fill in the specific information.