1
Foreword
In the long history of human beings fighting against infectious diseases, vaccines are a favorable weapon for human beings to fight infectious diseases. The accurate delivery of effective vaccines into the human body is the first step to complete the immune process. step. From the advent of subcutaneous injections in the mid-to-late 19th century, and later the advent of more advanced syringes, injections have become the main route of immunization today.
Injecting vaccines, however, is more demanding: vaccines are often formulated as unstable liquids that require refrigeration, or lyophilized powders for reconstitution; injections require trained technicians; needle sticks Injuries and needle re-use increase the potential for cross-contamination; finally, due to injection-related needle phobia and pain, especially paediatrics reduce injection vaccination compliance. Furthermore, the injection of the vaccine mainly induces a systemic immune response rather than targeting the infected area of the pathogen.
Most pathogens enter the body through mucosal surfaces, and nearly 80% of total immune cells in healthy adults are associated with mucosal surfaces. Different from injection immunization, mucosal immunization can not only induce systemic responses, but also well stimulate mucosal tissue-resident T cell responses and promote mucosal secretion of immunoglobulin A (IgA) antibodies. memory, so as to more effectively prevent the infection of mucosal pathogens. Mucosal vaccines also have the advantages of needle-free administration, such as lower risk of infection, higher compliance of vaccinators, and lower requirements for technicians.
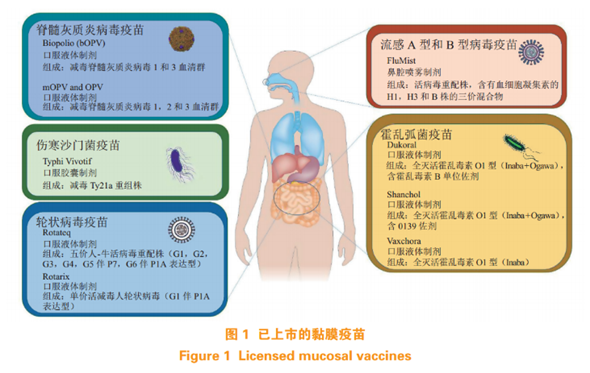
At present, although there are vaccines for intestinal and nasal mucosal immunization on the market, there are only a handful of them (4 types of intestinal mucosal and 1 type of nasal mucosal influenza vaccine, see Figure 1) . Mucosal vaccines also have their limitations. Most of these vaccines are attenuated or inactivated vaccines, and there are hidden dangers such as incomplete inactivation and virulence recovery.
In recent years, inhaled vaccines have become a hotspot in research and development in the prevention of respiratory microbial infections. The respiratory tract mucosa has a surface area of about 70-100 square meters, and has the characteristics of good epithelial permeability and high perfusion, and is one of the best targets for biological drug absorption. In particular, an inhaled new coronavirus vaccine in China has obtained an emergency use license, which once again focuses our attention in this direction. To study inhaled vaccines, the first step is to further deepen the understanding of respiratory mucosal immunity.
2
“Triple Protection”
Respiratory mucosal immune system
The respiratory mucosa is continuously exposed to foreign bodies for a long time. In the face of foreign bodies, whether it induces an immune response to protect the body or induces immune tolerance has established a balance mechanism. Typically, respiratory inhalation of foreign bodies such as dust particles and other inert substances is immune-tolerant, cleared by the mucociliary or by the cough/sneeze reflex.
However, when pathogens invade, the respiratory mucosa has a “triple” immune protection mechanism. The first is the conventional pathway: APC cells (antigen-presenting cells) recognize and absorb antigens through pathogen-associated molecular patterns (PAMPs) or damage-associated molecular patterns (DAMPs). Thereafter, DC cells (dendritic cells) typically migrate through afferent lymphatics to nearby draining lymph nodes, where antigens are presented to naive T and B cells via MHC-II complexes.
In addition to this regular set of antigen presentation pathways, there is a “second layer” of protection against human respiratory infection by the formation of specialized bronchial-associated lymphoid tissue (BALT) organize. The basic structure of BALT includes a central lymphoid follicle, overlying mucosal cells, and some variant epithelial cells. BALT can present antigens to naive T cells and effector T and B cells without migrating through the lymphatic system. There is a similar tissue near the nasal mucosa, the nasal-associated lymphoid tissue (NALT). BALT and NALT jointly participate in the mucosal immune system of the respiratory tract, and the elicited humoral immune response is very important for efficient processing of pathogenic antigens and subsequent pathogen neutralization. The humoral immune responses triggered by BALT are mainly local B cell responses and IgA responses. IgA antibodies in mucosal tissues are usually dimers, and secreted IgA (SIgA) is eventually formed, which can cross-link pathogens in the lumen, resulting in The steric barrier, thereby blocking infectivity, can also bind antigens within infected cells and expel them through vesicular transport.
The “third-level” protective mechanism of mucosal tissue is that antigen priming in one specific mucosal region induces a response in a different mucosal region. Briefly, antigen priming in the nasal or oral mucosa may induce mucosal antibodies in the mucosal tissue of the lung and vice versa. In other words, if a person is infected through the nasal mucosa, this may also lead to elevated SIgA levels in the airway mucosa. This interaction of different mucosal sites is often referred to as the common mucosal immune system.
3
Guanshan Difficulty Crossing
Hinders with inhalable vaccines
Structure determines properties, and the specific immune system of the respiratory mucosa determines its role in immunization. A successful inhaled vaccine needs to meet the following requirements: the vaccine can be delivered to the lungs accurately; sufficient immunogenic activity to ensure sufficient (mucosal) immune cell activation, i.e. it The immune tolerance barrier should be overcome; adverse reactions of the vaccine are few, especially not causing asthma and COPD. The following barriers must be overcome in vaccine development.
Moderate immune tolerance barrier
1
Since the respiratory mucosa needs to maintain a certain immune tolerance, it is ensured that hypersensitivity reactions will not occur during continuous exposure to a large number of harmless substances from the external environment. However, this “exquisite physiological design” has greatly tested the immunogenicity of the vaccine, so inhalable types often need to be used with adjuvants. The choice of adjuvant depends on the type of vaccine and the inherent immunogenicity of the vaccine itself. However, since the necessity of adjuvant in inhalable vaccine formulations has not been fully elucidated, the issue of immune response is considered in the study of inhaled vaccines. Immune cells and local antibody production, and to see if adjuvants increase the protective efficacy of the vaccine.
While the mucosal immune system plays a critical role in protecting the airways from invading pathogens, an excessive immune response can damage the lungs and lead to (antibody-dependent) respiratory disease. Several studies have shown that, in some cases, an excessive immune-related response leads to the development of cytokine storms.
Particle size and deposition
2
Since the area of the respiratory tract infected by airborne pathogens depends on the expression of their respective attachment receptors, the deposition site of inhaled vaccines is associated with the exertion of vaccine effects, so vaccine formulations should also be considered Disperse within the aerodynamic size range and select the particle size suitable for penetrating the airway. Typically 1-5 µm aerosols are suitable for penetration and deposition in the lungs, while smaller particles are easily exhaled. Particles larger than 5 µm are mainly deposited in the throat or upper respiratory tract. In addition to particle size, other factors such as shape, density, inhalation flow rate, electrical charge, and hygroscopicity may also affect particle deposition behavior in the lungs.
The Dilemma of Modeling
3
A prerequisite for in vivo studies is that vaccines should be rigorously evaluated in animal models closest to humans. In vivo studies of vaccines often require animal models, but they cannot be performed desired inhalation action.
In addition, the patient’s inhalation behavior is a factor that affects the effectiveness of inhaled vaccines. The patient can perform the inhalation action correctly so that the vaccine can reach the desired target area. To maintain the appropriate breathing flow rate and the correct hand-breathing coordination, all these require the correct inhalation operation guidance. In many trials, the use of dry powder inhaler (DPI), which is a relatively mature inhalation device, is considered to optimize the deposition of vaccine in the lungs with reference to dry powder inhalation, and a special valve is introduced to facilitate the use of children .
Applicability Considerations
4
Inhalable vaccine formulations can be broadly divided into two categories: liquid formulations and powder formulations. Dry powder formulations have several advantages over liquid formulations. First, properly dried vaccines can be more stable and easy to store in ambient conditions, thus avoiding cold chain requirements. Second, the dry vaccine weight is smaller than the liquid solution, which is convenient for bulk transportation; third, the dry powder formulation can be adapted to a disposable dry powder inhaler, which can prevent repeated use, cross-contamination and degradation caused by moisture, and compared with liquid formulations, it can be Dose control during inhalation. For example, Keweisha ®Fogyou® uses a nebulizer to atomize the vaccine of dry powder preparation into tiny particles, and complete the vaccination by oral inhalation.
4
Successful example
Live attenuated influenza vaccine (LAIV)—FluMist, manufactured by AstraZeneca and approved by the FDA in 2003, is the most successful nasal vaccine to date.
FluMist overcomes the barriers of low nasal mucosal permeability, high mucociliary clearance, mucus barrier and the enzymatic environment of the nasal mucosa and has been used in children and adults for over 10 years. The vaccine contains three attenuated virus vaccines: two type A strains (H1N1 and H3N2 subtypes) and one type B strain. Each virus has an identical backbone consisting of six gene segments from the main donor virus and two gene segments from wild-type influenza virus (encoding the hemagglutinin and neuraminidase glycoproteins), and each LAIV virus has is cultured and purified from eggs. Through genetic engineering, the live attenuated virus infects and replicates in nasopharyngeal mucosal cells, but does not invade the lower respiratory tract and lungs. The ability of live attenuated virus to infect and replicate in the nasopharynx enhances antigen capture and presentation, favoring the development of an immune response. Furthermore, this delivery system allows the antigen to be presented in its native form, and thus induces an immune response similar to that induced by natural infection.
The preparation contains some trace impurities that act as adjuvants. All these features and their good security features are the reasons for FluMist’s success. In February 2012, the FDA approved FluMist quadrivalent, a formulation containing two influenza A subtype viruses and two influenza B viruses, and the use of the population has also expanded to the population of 2 to 49 years old
5
Summary
Among all viral respiratory infections, the only “routinely available” inhaled vaccine is currently against the influenza virus. In the current COVID-19 pandemic, inhaled vaccines are a potential way of administration, especially dry powder vaccines can improve stable and efficient vaccination. Compared with injectable vaccines, inhaled vaccines have a wider application area, are more friendly to the elderly, weak and young, and are safer. In addition, the stronger mucosal immune response induced by inhaled vaccines may provide an additional line of defense. It is hoped that inhaled vaccines can enrich the arsenal and fight respiratory diseases more effectively.
Main References
[1]Sun Xun, Zhang Yuandong. Research progress of mucosal vaccine delivery technology to overcome immune barrier[J]. Advances in Pharmacy, 2022,46(4):282-295.
[2]Mato Y L. Nasal route for vaccine and drug delivery: Features and current opportunities[J]. International Journal of Pharmaceutics, 2019,572:118813.
[3]Heida R, Hinrichs W L, Frijlink H W. Inhaledvaccine delivery in the combat against respiratory viruses: a 2021 overview of recent developments and implications for COVID-19.: REVIEW[J]. Expert Review of Vaccines, 2021.
[4]Benne N, Duijn J V, Kuiper J, et al. Orchestrating immune responses: How size, shape and rigidity affect the immunogenicity of particulate vaccines[J]. Journal of Controlled Release, 2016,234:124-134.