Recently, Henan Real Biotechnology Co., Ltd. (hereinafter referred to as “Real Bio”) announced that the clinical trial application (IND) of the self-developed new generation oral long-acting HIV drug candidate CL-197 will be filed in 2022 On October 8, 2018, it was officially approved by the Center for Drug Evaluation (CDE) of the National Medical Products Administration (NMPA).

Picture: from CDE official announcement
CL-197 is a novel nucleoside reverse transcriptase inhibitor NRTI (Nucleoside Reverse Transcriptase Inhibitor), which can inhibit by mimicking (ie competing with) endogenous purine nucleotides Reverse transcription, with a potential long-acting role in the treatment of HIV infection. The preclinical study completed in December 2021 showed that CL-197 can effectively inhibit the replication of HIV, and it is expected to become a long-acting AIDS treatment drug that only needs to be taken orally once a week.
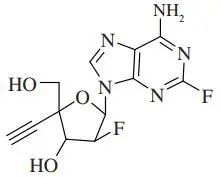
Figure: Chemical formula of CL-197
So far, there are more than 41 million AIDS patients worldwide. The current treatment plan is limited by “daily medication, drug toxicity, high treatment cost, and drug resistance”. Sex has plagued many AIDS patients. In this regard, Dr. Du Jinfa, Executive Director, Chairman, CEO and CSO of Real Bio, said that CL-197, as a new generation of oral long-acting HIV treatment candidate drug, has 3 bright spots: (1) Effective (2) Oral long-acting characteristics: pharmacokinetic studies show that the half-life of the active ingredient of CL-197 in PBMC is about 168 hours; (3) It is expected to be administered orally once a week: a more convenient dosing regimen Will increase medication adherence and improve clinical outcomes. In addition, the company’s AIDS treatment drug Azvudine, which will be approved for marketing in 2021, has dual-target and long-acting characteristics. The combination of CL-197 and Azvudine meets the elements of a complete AIDS cocktail therapy. It is equivalent to three drugs with three mechanisms of action acting at the same time, which can effectively prevent the occurrence of drug resistance. It is expected to be developed into the world’s first all-oral long-acting drug AIDS treatment medicine.
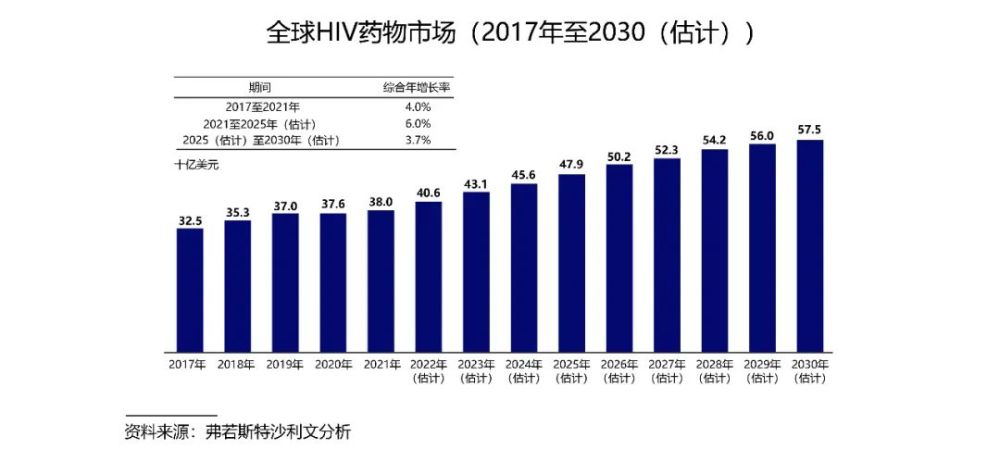
Current HIV drug development focuses on improving drug safety, efficacy, drug resistance, and simplifying treatment to improve drug adherence. As the backbone components of all first-line antiretroviral therapy recommended by WHO for HIV-infected patients, nucleosides are an important part of the HIV drug market and have enormous growth potential. According to Frost & Sullivan, the global HIV drug market has been growing steadily, from US$38 billion in 2021 to US$57.5 billion in 2030.
After this IND approval, True Bio will conduct a single-center, randomized, double-blind, placebo-controlled and single ascending dose trial to evaluate the efficacy of CL-197 in healthy subjects Safety, tolerability and pharmacokinetic characteristics in subjects. As part of the global expansion strategy, the company also plans to apply for IND in overseas jurisdictions after applying for IND in China, aiming to improve R&D efficiency and benefit patients around the world as soon as possible.
About real creatures
Real Bio is an R&D-driven biotechnology company dedicated to the development, manufacture and commercialization of innovative drugs for the treatment of viral, oncology and cerebrovascular diseases. The company’s mission is to improve human health through true innovation. Through the unremitting pursuit of innovation, we aim to find newer and better treatment solutions to meet the unmet clinical needs, and gradually establish the advantages of real biology in the research and development of antiviral and antitumor drugs, and become a globally competitive company. biopharmaceutical company.