▎WuXi AppTec Content Team Editor
A few days ago, researchers at Calibr, part of the Scripps Research Institute, announced the results of the first human clinical trial of its controllable CAR-T cell therapy. The uniqueness of this technology is that the activity of CAR-T cell therapy can be turned on or off through antibody therapy, thereby making CAR-T cell therapy safer and more effective. Preliminary results from the first human trial show that this controllable CAR-T cell therapy not only showed a 67% complete response rate in the treatment of B-cell blood cancer patients who had received various previous treatments , and shorten the duration of adverse reactions to 2-3 days, which is significantly shorter than the approved CAR-T therapy (5-17 days).
CAR-T therapy has shown excellent efficacy in the treatment of blood cancers, and a number of therapies have been approved for marketing. However, these CAR-T cells targeting tumor antigens may produce cytokine release syndrome (CRS) and neurotoxicity after activation. These adverse reactions, if not controlled, may lead to the death of patients in severe cases.
These adverse reactions occur because after CAR-T cells are activated, they continue to activate, triggering a broad inflammatory response. Therefore, researchers have been exploring whether to add a switch to CAR-T cells to instantly regulate the activity of CAR-T cells.
This is what Calibr researchers are working on. The controllable CAR-T therapy they designed consists of two components. The chimeric antigen receptor expressed on CAR-T cells named CLBR001 does not target the antigen expressed on the surface of tumor cells, but targets a adaptor protein. This protein is not present in humans, so they are not activated when CLBR001 is infused into humans. Another component of this therapy, SWI019, is a fusion protein targeting the CD19 antigen. One end of it can recognize the CD19 antigen expressed by tumor cells, and the other end can bind to the CAR expressed by CLBR001. When SWI019 was also infused into patients, it combined with CLBR001 to target CD19 and activated CAR-T cells to destroy tumors.
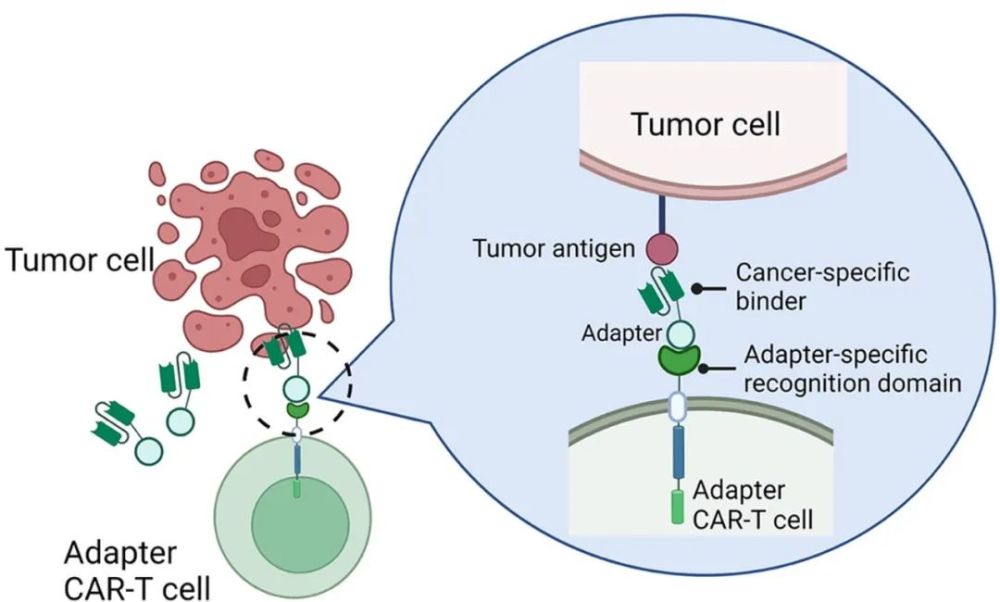
▲The mechanism of action of controllable CAR-T therapy (Image source: Reference [3])
The advantage of this therapy is thatSWI019 has a short half-life, so it is degraded after CAR-T cell activation, thereby shutting down CAR-T cell activity . Safety data from the clinical trial released today also showed that only two of the nine patients treated experienced grade 3 or higher CRS or neurotoxicity. Symptoms of CRS or neurotoxicity of any grade can be eliminated in only 2-3 days, while these adverse reactions of approved CAR-T therapy are eliminated in 5-17 days.
In terms of efficacy, 7 of the 9 treated patients achieved remission, including 6 complete remissions. These preliminary efficacy data show the efficacy of this controllable CAR-T therapy is similar to the currently approved CAR-T therapy.
Another advantage of this controllable CAR-T therapy is that as long as SWI019 is injected again, the activity of CAR-T cells can be reactivated. Moreover, if the patient’s tumor becomes resistant to CD19-targeting CAR-T therapy, researchers can enter fusion proteins targeting other antigens, allowing CLBR001 to target other antigens, which is expected to overcome tumor resistance.
“These results show the potential of Calibr’s controllable CAR-T platform. CLBR001 is like the hardware of a computer, and a protein with a switch function is like software that can program a computer.” Calibr Biologics “This universal platform can be programmed to target any target, including solid tumor targets. Today’s data are just the first step toward demonstrating its potential,” said Dr. Travis Young, Vice President.
As WuXi AppTec’s CTDMO focused on cell and gene therapy, WuXi Bioscience is committed to accelerating and transforming the development, testing, manufacturing and commercialization of gene and cell therapy and other high-end treatments. WuXiXi can help global customers bring more innovative therapies to the market as soon as possible for the benefit of patients. If you have relevant business needs, please click the picture below to fill in the specific information.